“I am always doing that which I cannot do, in order that I may learn how to do it.” Pablo Picasso
“We have more possibilities available in each moment than we realize.” Nhat Hanh
Précis: This post exists because I received a note from a reader concerned about using a substance that potentially was an immune enhancer, coupled with the notion that Parkinson’s was an autoimmune disease. And to top it off, one of the papers in a pile of papers on my desk was a recent paper concerning memory T-cells (lymphocytes) that had Parkinson’s-specific gene signatures. Thus, this post will review the evidence that supports Parkinson’s being classified as an autoimmune disease.
Note: If you want to distill this information into a 1-page handout on “Parkinson’s and Autoimmunity,” just click on the link below.
“To raise new questions, new possibilities, to regard old problems from a new angle, requires creative imagination and marks real advance in science.” Albert Einstein
Then and Now: As a long-time research scientist, I spent much of my career reading, pondering, reading more, and thinking even more. And then, we would formulate a testable hypothesis from the information in these readings and propose and design experiments (some of these studies would take 3-4 years). Preliminary results would lead to research proposals, and funded studies would advance our work. Then it would start all over, read, write, design experiments, and more grant proposal writing. But that was then.
Today, I read old and new papers on various areas of science and a lot about Parkinson’s. But now I formulate more of an opinion and not so much a hypothesis. Furthermore, instead of designing an experiment or a study, I do my best to translate the science and medicine such that everyone can read it and better appreciate/understand the details of this work.
But the topic of this blog post is the immune system, which is enormous and rich with complexities and nuances. I re-read some foundational review papers published >20 years ago. They are still relevant and entirely meaningful today. So, coupled with some newer articles on the immune system related to Parkinson’s and the central nervous system, my brain says, let’s do it.
Some classic review articles about the immune system-
•Davidson, Anne, and Betty Diamond. “Autoimmune diseases.” New England Journal of Medicine 345, no. 5 (2001): 340-350.
•Marrack, Philippa, John Kappler, and Brian L. Kotzin. “Autoimmune disease: why and where it occurs.” Nature medicine 7, no. 8 (2001): 899-905.
•Delves, Peter J., and Ivan M. Roitt. “The immune system.” New England journal of medicine 343, no. 1 (2000): 37-49.
•Parkin, Jacqueline, and Bryony Cohen. “An overview of the immune system.” The Lancet 357, no. 9270 (2001): 1777-1789.
A few recent review articles about the immune system and Parkinson’s and the CNS-
•Sabatino Jr, Joseph J., Anne-Katrin Pröbstel, and Scott S. Zamvil. “B cells in autoimmune and neurodegenerative central nervous system diseases.” Nature Reviews Neuroscience 20, no. 12 (2019): 728-745.
•Jain, Rajiv W., and V. Wee Yong. “B cells in central nervous system disease: diversity, locations and pathophysiology.” Nature Reviews Immunology 22, no. 8 (2022): 513-524.
•Harms, Ashley S., Sara A. Ferreira, and Marina Romero-Ramos. “Periphery and brain, innate and adaptive immunity in Parkinson’s disease.” Acta Neuropathologica 141, no. 4 (2021): 527-545.
•Mayne, Katie, Jessica A. White, Christopher E. McMurran, Francisco J. Rivera, and Alerie G. de la Fuente. “Aging and neurodegenerative disease: is the adaptive immune system a friend or foe?.” Frontiers in aging neuroscience 12 (2020): 572090.
“Sometimes I’ve believed as many as six impossible things before breakfast.” Lewis Carroll
What is the Definition of an Autoimmune Disease? Davidson and Diamond (2001) “defined an autoimmune disease as a clinical syndrome caused by the activation of T cells or B cells, or both, in the absence of an ongoing infection or other discernible cause.” Thus, an autoimmune disease is the inappropriate activation of the host’s immune system to attack self-antigens (that is, you).
“I love those who yearn for the impossible.” Johann Wolfgang von Goethe
Important Terms/Definitions
•Self-antigens: an individual’s own antigens
•Immunologic tolerance: unresponsiveness to self-antigens
•Tolerogens: antigens that induce tolerance
•Tolerance: the prevention of an immune response against a particular antigen. For instance, the immune system is generally tolerant of self-antigens, so it does not usually attack the body’s own cells, tissues, and organs
•Immunogens: antigens that induce an immune response
•Immunity: resistance to infectious disease and the collection of cells and tissues that protects the body from infection is known as the immune system. The coordinated reaction of the cells of the immune system to a pathogen is known as the immune response.
•Autoimmunity: failure of self-tolerance and resulting immune reaction to self-antigens
“The only limits to the possibilities in your life tomorrow are the buts you use today.” Les Brown
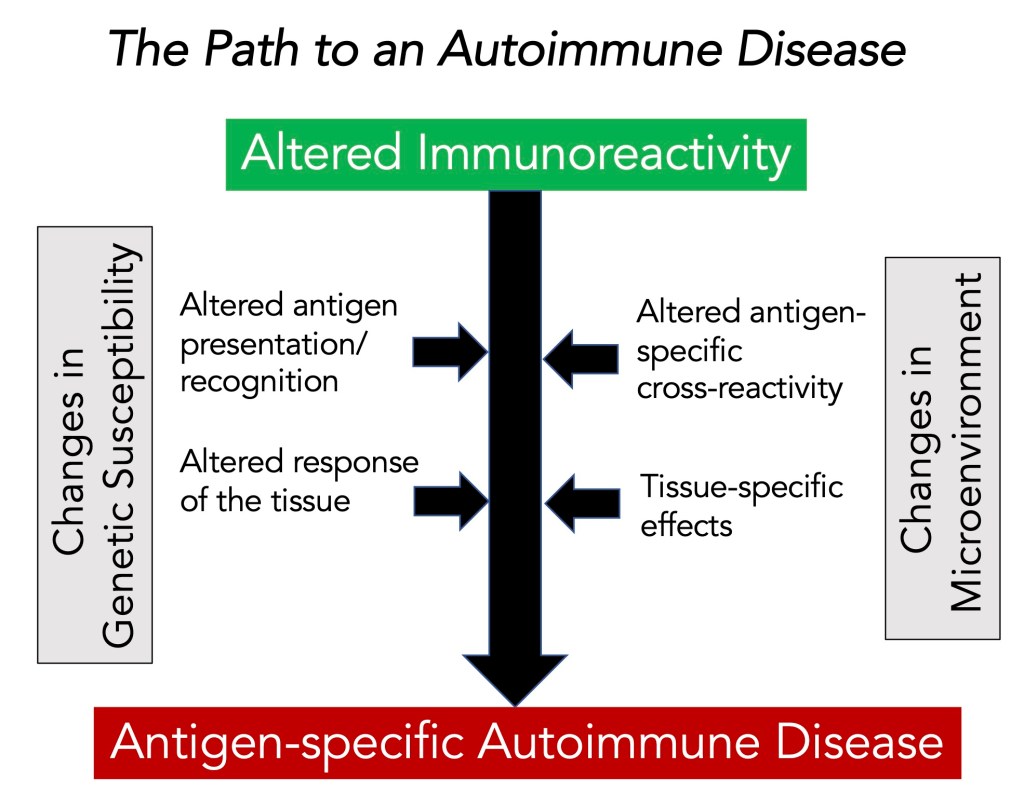
Tolerance and Autoimmunity: In past work, we said, “The immune system is a complex interacting network of organs and tissues, cells, and molecules designed to work together to identify and protect the body from infectious pathogens and other diseases.” Our immune system can respond to billions of foreign antigens without being exposed to antigens. Since the structure of a foreign antigen and self-antigens (or autoantigens) are essentially the same, our immune system is programmed to respond to antigens only under highly regulated and specific microenvironments (usually in the presence of inflammatory cytokines).
An important concept is that immunological self-tolerance is unresponsiveness to specific antigens induced by exposure of lymphocytes to that antigen. For example, central tolerance induced by immature lymphocytes encountering antigens in generative, central lymphoid organs leads to negative selection in the thymus for T cells and bone marrow for B cells, and the development of regulatory T cells. Peripheral tolerance induced by mature lymphocytes encountering antigens in peripheral tissues leads to T and B cell deletion, suppression of that clone, and anergy (a condition in which the body fails to react to an antigen).
Unfortunately, we do not have a foolproof safety system for tolerance of ‘self.’ Autoimmune disease results when a breakdown occurs in the mechanism to preserve tolerance. About 5% of Americans suffer from some form of autoimmune disease. Most autoimmune diseases occur when the layers of tolerance-inducing processes fail to eliminate self-reactive cells in genetically normal individuals.
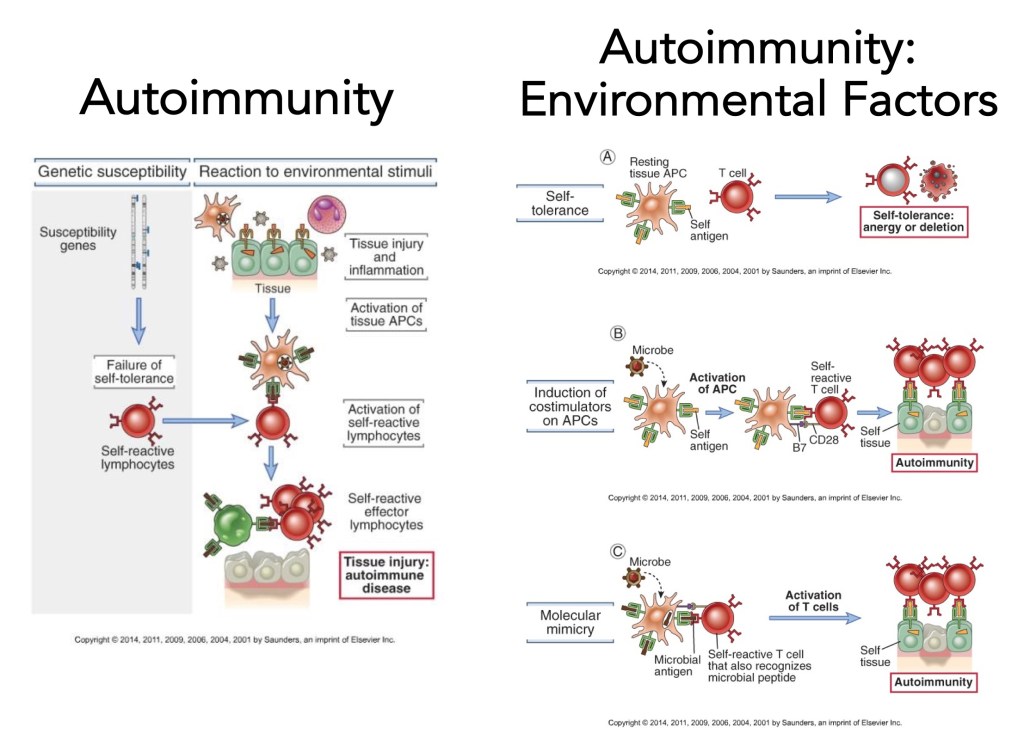
Three Requirements for Autoimmunity to Occur-
•First, an individual must express MHC molecules that efficiently present a peptide derived from the target self-antigen. [The major histocompatibility complex (MHC) of genes consists of a linked set of genetic loci encoding many of the proteins involved in antigen presentation to T cells, most notably the MHC class I and class II glycoproteins.] In other words, the MHC molecules you inherit play a role in determining your susceptibility to an autoimmune disease. For example, only ~0.2% of the U.S. population suffers from juvenile diabetes; however, they inherit two particular types of class II MHC, and the probability of contracting juvenile diabetes increases ~20-fold.
•Second, an individual must produce T and, in some cases, B cells with receptors that recognize a self-antigen. A mix-and-match strategy makes our immunologic diversity so that everyone is different from everyone else. Thus, by chance, a person will produce lymphocytes whose receptors recognize a self-antigen.
•Third, environmental factors must lead to the breakdown of the tolerance mechanisms, which have been designed to eliminate self-reactive lymphocytes. The current favorite hypothesis is that microbial attack (bacterial or viral) triggers the event as a ‘last straw’ that leads to autoimmune disease- we call it “molecular mimicry.” The schematic below illustrates some of these ‘issues.’
“When you have exhausted all possibilities, remember this – you haven’t.” Thomas A. Edison
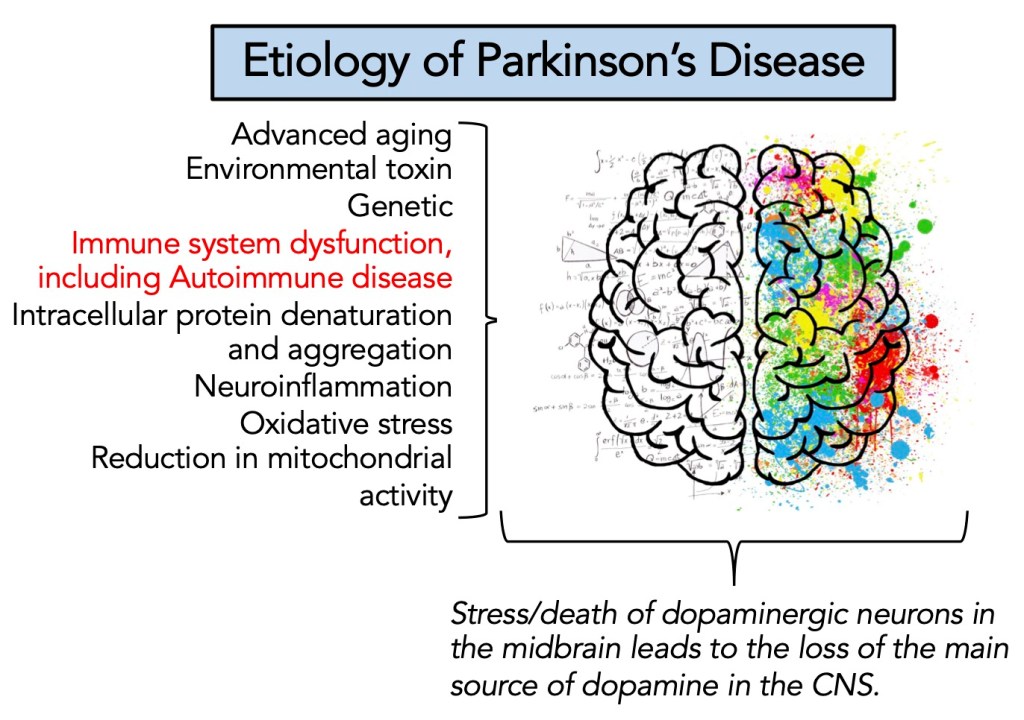
Abnormal Immune Response Promotes the Development of Parkinson’s: The occurrence of intracellular inclusions named Lewy bodies is associated with the development of Parkinson’s. A major component of Lewy bodies is aggregates of the protein alpha-synuclein (αSYN). A possible scenario leading to Parkinson’s in older adults includes αSYN aggregation in dopaminergic neurons. The response to αSYN aggregation is cytokine-driven neuroinflammation, which enables an age-linked immunologic dysfunction. There is also evidence that αSYN, usually an intracellular protein, becomes an autoantigen released into the extracellular neuronal cell spaces and then aggregates, further activating the immune response to engage the now deranged neuronal cells. Thus, over time, the afflicted neuron becomes dysfunctional and continues to be engaged by the inflammatory and immunological systems; dopaminergic neurons are destroyed, and Parkinson’s begins.
“nothing is impossible” Walt Disney
Highlights From Recent Papers That Support Parkinson’s to be an Autoimmune Disease: What has caught my attention is derived from these papers, all exciting reading.
Jiang, Tianfang, Gen Li, Jun Xu, Shane Gao, and Xu Chen. “The challenge of the pathogenesis of Parkinson’s disease: Is autoimmunity the culprit?.” Frontiers in immunology 9 (2018): 2047.
•The overarching theme by Jiang et al. (2018) is that autoimmunity has a significant role in the pathogenesis of Parkinson’s, including a descriptive role for several proteins a-synuclein, DJ-1, PINK1, and Parkin. Importantly, PINK1, Parkin, and DJ-1 are neuroprotective when expressed. By contrast, deficiencies of these genes promote a hyperreactive immune reaction. The initiator role of a-synuclein to initiate the autoimmune response is reinforced here. The evidence of molecular mimicry is reviewed between herpes simplex virus-1 and human a-syn, further supporting the role of a-SYN in the autoimmune process. The part of immune cells in the autoimmune process for T cells, B cells, dendritic cells, and microglia cells is further discussed.
Bonam, Srinivasa Reddy, and Sylviane Muller. “Parkinson’s disease is an autoimmune disease: A reappraisal.” Autoimmunity Reviews 19, no. 12 (2020): 102684.
•Bonam and Muller (2020) briefly reinforce the immunity link in familial forms of Parkisnon’s, including LRRK2 (DARDARIN; leucine-rich repeat kinase 2/PARK8), PRKN (PARKIN/E3 ubiquitin ligase/PARK2), PINK1 (PTEN-induced putative kinase/PARK6), and J-1 (DJ1 protein/PARK7) have been mainly investigated in addition to a-SYN Furthermore, they summarize the evidence of autoantibodies having been detected in the serum from Parkinson’s patients. These antibodies cross-react with neuronal antigens and neuronal cells. Finally, they discuss the T cell response to Parkinson’s disease-related antigens, ultimately suggesting that a-syn-reactive T cells may have some predictive value in the disorder. This review supports the notion of autoreactive T cells and B cells in the pathogenesis of Parkinson’s.
Gate, David. “New perspectives on immune involvement in Parkinson’s disease pathogenesis.” Journal of Parkinson’s disease Preprint (2022): 1-7.
•Gate (2022) nicely summarizes the immune system’s role in the pathogenesis of Parkinson’s. Included is the dysfunctional part of humoral and cellular immunity, with the role of genetics and epidemiological studies pointing to autoimmune disorders. Interestingly, autoimmune diseases and Parkinson’s share common genetic paths. Many loci have been identified between Parkinson’s and type I diabetes, multiple sclerosis, Crohn’s disease, celiac disease, psoriasis, and ulcerative colitis. Identifying these shared and common pathways further argues for the importance of immunogenetics in Parkinson’s and potential immune therapy.
Tansey, Malú Gámez, Rebecca L. Wallings, Madelyn C. Houser, Mary K. Herrick, Cody E. Keating, and Valerie Joers. “Inflammation and immune dysfunction in Parkinson disease.” Nature Reviews Immunology (2022): 1-17.
•If I was asked to give a gold star to one paper here, it would be given to Tansey et al. (2022). They expertly discuss the importance of the inflammatory and immune systems in the cause of Parkinson’s. They review markers of inflammation and immune cells in the blood and spinal fluid of Parkinson’s patients. Furthermore, they highlight several key risk factors, combined with disease genes, that modify the immune function in Parkinson’s, which support a role for viral or bacterial infection, pesticides, and gut microbiota in promoting Parkinson’s. The dysfunction found in an aging immune system, coupled with “complex gene-by-environment,” provides an appropriate initiation process that leads to Parkinson’s. Ultimately, the long-term goal and hope are for earlier detection of the disorder, preventing or delaying progression, and effectively treating this disease.
Xing, Frank, Luca Marsili, and Daniel D. Truong. “Parkinsonism in viral, paraneoplastic, and autoimmune diseases.” Journal of the Neurological Sciences 433 (2022): 120014.
•Xing et al. (2022) focus on secondary parkinsonism, which is parkinsonism besides classical idiopathic neurodegeneration. Secondary causes of parkinsonism, such as infectious (mainly viral and prion-like diseases), autoimmune (systemic/drug-induced), and paraneoplastic etiologies, are rare but are a topic of increasing interest. Due to the COVID-19 pandemic, there is renewed interest in the dual hit hypothesis of Parkinson’s, which suggests that a viral infection may collaborate and worsen cellular degradation in already ill patients, which could favor more significant amounts of denatured and secreted a-SYN. Along these same lines, dysfunctional parts of the immune response may promote the production of autoantibodies to the neuronal cell surfaces.
Dhanwani, Rekha, João Rodrigues Lima-Junior, Ashu Sethi, John Pham, Gregory Williams, April Frazier, Yaqian Xu et al. “Transcriptional analysis of peripheral memory T cells reveals Parkinson’s disease-specific gene signatures.” npj Parkinson’s Disease 8, no. 1 (2022): 1-10.
•Dhanwani et al. (2022) strengthened the findings of Parkinson’s-associated autoimmune disorders characteristics for the pathogenesis of Parkinson’s. They began by studying RNA sequencing on blood monocytes and peripheral CD4 and CD8 memory T cell subsets of cells isolated from patients with Parkinson’s. They used the response to a-SYN as the insulting proinflammatory response, revealing a Parkinson’s-specific gene expression profile in the memory T cell subsets. The enrichment profiles were in oxidative stress, phosphorylation, autophagy of mitochondria, cholesterol metabolism, inflammation, and several chemokine signaling proteins. By studying the peripheral blood cells, they noted genes expressed previously and found in neurons, including LRRK2, LAMPS3, and aquaporin. These exciting results suggest a substantial autoimmune disease linked to the pathogenesis of Parkinson’s.
“All things are possible until they are proved impossible – and even the impossible may only be so, as of now.”
Linking Autoimmunity to Parkinson’s: Neuroinflammation is related to the etiology of Parkinson’s. Now we link autoimmunity. It began with the epidemiological linkage of common genetic loci between Parkinson’s and autoimmune diseases. They have found that patients with multiple sclerosis, Graves’ disease, Hashimoto disease, and pernicious anemia have an increased risk of developing Parkinson’s. Recently, Genome-wide association studies (GWAS) found seventeen shared loci between autoimmune diseases and Parkinson’s; this includes type 1 diabetes, Crohn’s disease, ulcerative colitis, coeliac disease, psoriasis, rheumatoid arthritis, and multiple sclerosis.
“I don’t need easy, I just need possible.” Bethany Hamilton
Summary: These results suggest a group of autoimmune disorders is linked to Parkinson’s. The peripheral immune system is chronically turned-on and able to actively produce inflammatory mediators that fuel the neuroinflammatory flame that promotes the pathogenesis of Parkinson’s. However, we need a more in-depth understanding of this immunological process and mechanism that links Parkinson’s to autoimmune diseases. From this knowledge, novel immunomodulation therapies could evolve to slow or delay the progression of Parkinson’s hopefully.
“Man often becomes what he believes himself to be. If I keep on saying to myself that I cannot do a certain thing, it is possible that I may end by really becoming incapable of doing it. On the contrary, if I have the belief that I can do it, I shall surely acquire the capacity to do it even if I may not have it at the beginning.” Mahatma Gandhi
Cover Photo Image by Jaesung An from Pixabay








One Reply to “”