“Come grow old with me. The best is yet to be.” William Wordsworth
“As long as I am breathing, in my eyes, I am just beginning.” Criss Jami
Introduction: Recently, an exciting and in-depth review article about inflammation and the immune system and their dysfunction in the pathway of Parkinson’s was published. The citation for this paper is as follows: Tansey, Malú Gámez, Rebecca L. Wallings, Madelyn C. Houser, Mary K. Herrick, Cody E. Keating, and Valerie Joers. “Inflammation and immune dysfunction in Parkinson disease.” Nature Reviews Immunology (2022): 1-17.
Due to the complex story told by this review article, I thought to present an overview of innate and adaptive immunity (Part 1), the changes found in aging adults (Part 2), and the changes found in aging adults with Parkinson’s (Part 3). However, simplifying the immune system is hard, and it almost defeats the purpose because understanding the little and big picture of immunity showcases how remarkable our bodies are in defending against pathogens.
If you are comfortable with your understanding of the immune system, skip down to the aging immune system section 2 or even the final section 3 on the aging immune system and how it contributes to the development of Parkinson’s.
Here is the content of the material presented below as an Introduction to the Immune and Inflammatory Systems [please remember, “the devil is in the details,” (that is, the details of the matter are its most problematic aspect) and believe me when I say, I left out most of the detail!]:
Immune and Inflammatory Systems
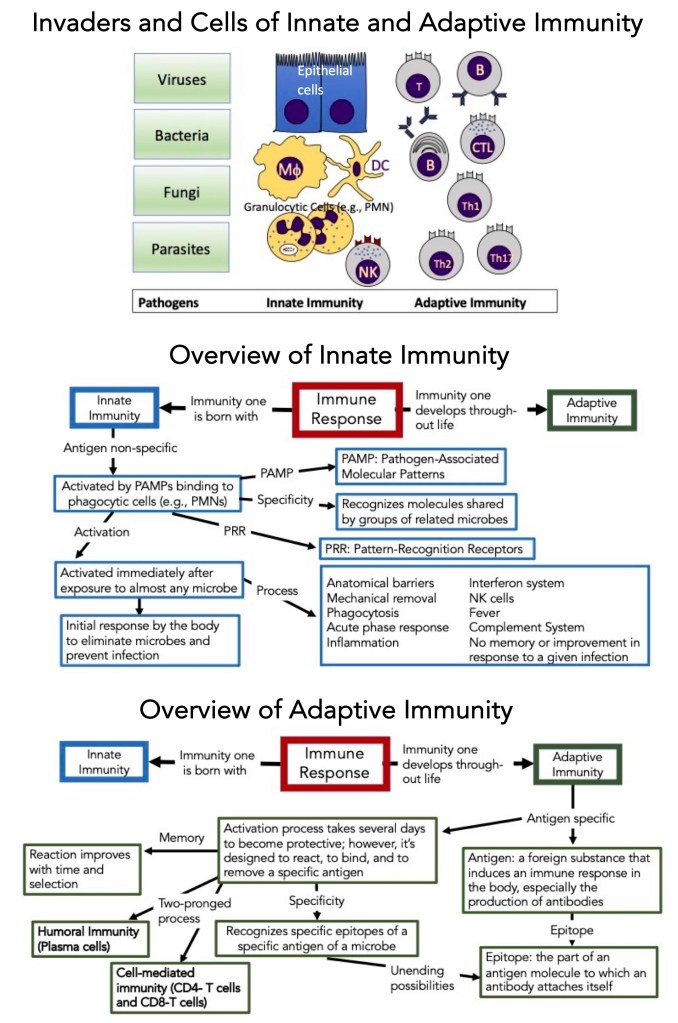
Invaders and Cells of the Innate and Adaptive Immune Systems
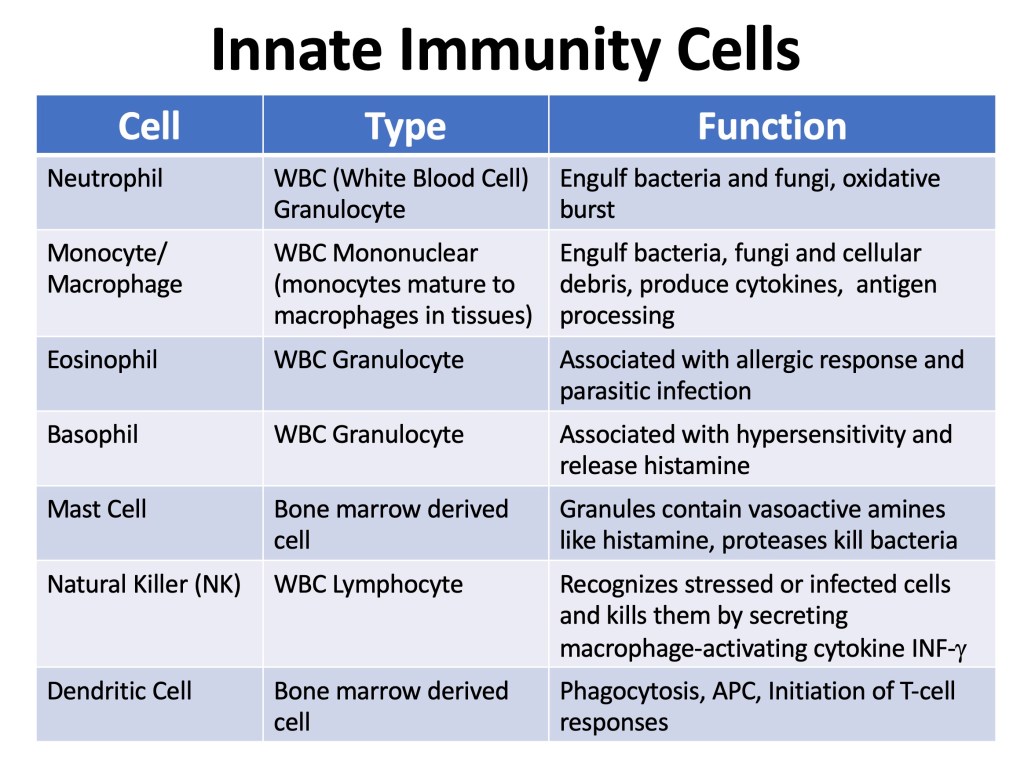
Innate Immunity Cells
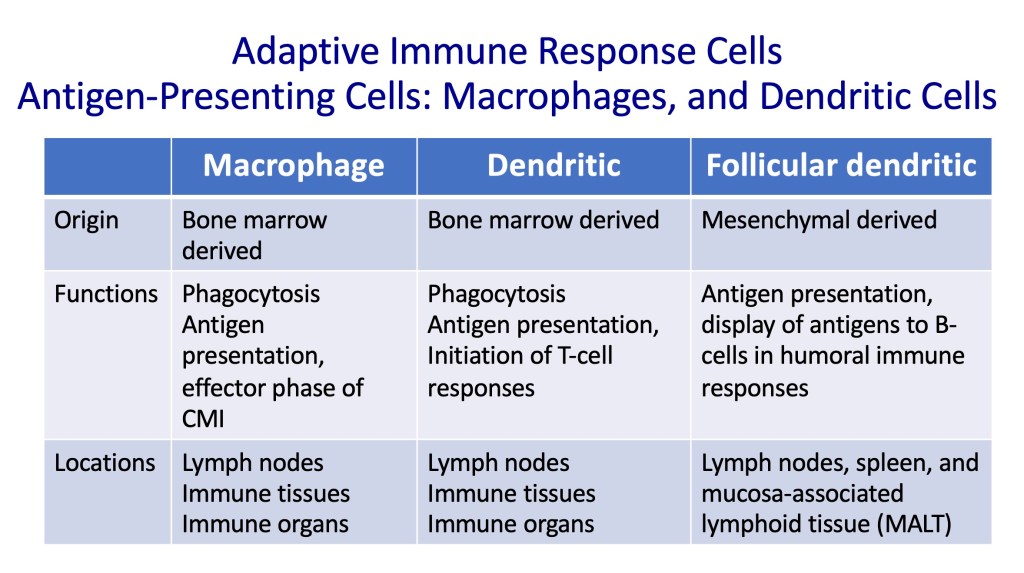
Adaptive Immunity Cells
Cytokines, the Regulators of Immunity and Inflammation
Examples of Important Cytokines
Uniting the Immune and Inflammatory Systems
Basic Vocabulary in Immunology
Organs and Tissues of the Immune System
Differences Between Acute Inflammation versus Chronic Inflammation
Phagocytic Cell Respiratory or Oxidative Burst: Order of Events
Pattern Recognition Receptors [Toll-like Receptors (TLR-1 through TLR-9)]
The Type I Interferon System of Innate Immunity
Defining Adaptive Immunity
Example of a Pro-Inflammatory Response is when Adaptive Immune Cells and Macrophages Encounter a Pathogen
“The idea is to die young as late as possible.” Ashley Montague
PART 1- The Immune and Inflammatory Systems: In writing about the immune system, COVID-19, and Parkinson’s, we said the following about the immune system (papers listed at the end of the blog post), “The immune system is a complex interacting network of organs and tissues, cells, and molecules designed to work together to identify and protect the body from infectious pathogens and other diseases.”
•Always remember that the most critical physiologic functions of the immune system are preventing infections and eradicating established infections.
•The first defense against microorganisms is the intact skin and mucous membranes.
•If microorganisms breach this line and enter the body, the immune system’s innate (natural) arm (second line of defense) is available to destroy the invaders.
•The first and second lines of defense cannot modify their response to pathogens.
•The third level of defense is specific protection provided by the immune system’s adaptive (acquired) arm; however, it takes several days for this defensive stage to fully function.
•The two components of the adaptive arm are cell-mediated immunity and antibody-mediated (humoral) immunity.
•It is essential to stress that all three levels of defense are critical for maintaining our health.
Here in the schematic form, are the details of the innate and adaptive immune systems.
The details of the cells involved in innate and adaptive immune systems are in table format below.

“Grow old with me! The best is yet to be.” Robert Browning
Cytokines Directing Cellular Traffic in Inflammation and Immunity: We previously summarized this information about cytokines and inflammation: “The immune system’s response to infection, injury, and toxic compounds is inflammation. The inflammatory process is a host-defense response mechanism. Cytokines orchestrate many of the immune and inflammation reactions.”
•Cytokines are low molecular weight, soluble proteins produced in response to an antigen and function as chemical messengers for regulating the innate and adaptive immune systems.
Cytokines are produced by virtually all cells involved in innate and adaptive immunity, especially by T helper lymphocytes.
•The activation of cytokine-producing cells triggers them to synthesize and secrete their cytokines.
•In turn, the cytokines can bind to specific cytokine receptors on other immune system cells and influence their activity.
•Uncontrolled inflammation can lead to host morbidity and mortality.
“There is no old age. There is, as there always was, just you.” Carol Grace
Combining Innate and Adaptive Immune Systems is Powerful: In previous work on the immune system, exercise, and Parkinson’s, we said, “The immune system protects the body against infection and non-native substances through intricate pathways that ultimately distinguish everything foreign from non-foreign within a given person.” The schematic below does an excellent job of visualizing the interplay of innate and adaptive immunity, and it illustrates the links between inflammation and the immune system given by acute inflammation and chronic inflammation ( image credit: Creative-diagnostics.com)
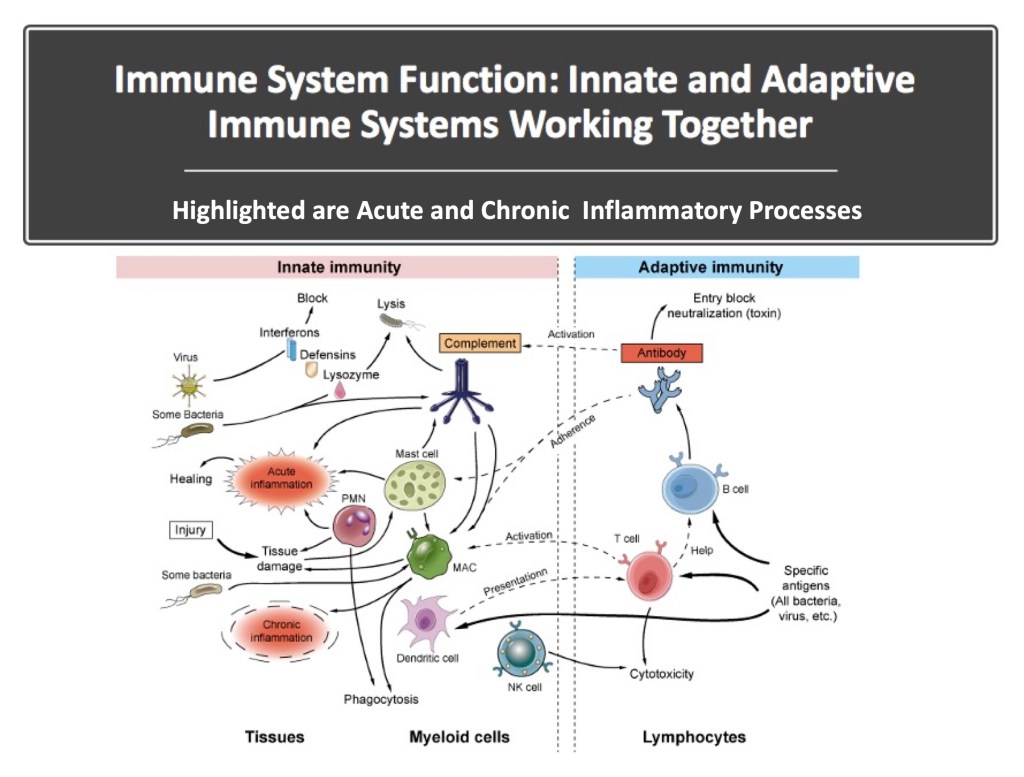
Basic Vocabulary in Immunology:
•Immunity is defined as resistance to infectious disease and the collection of cells and tissues that protects the body from infection is known as the immune system. The coordinated reaction of the cells of the immune system to a pathogen is known as the immune response.
•When B and T cells become activated, they divide and mature into effector cells that actually do the job of fighting the microbe. Mature B cells are called plasma cells; plasma cells secrete antibodies. Mature T cells are called effector T cells. Effector T cells either assist (“help”) leukocytes to kill ingested microbes or directly kill infected cells.
•Humoral immunity is the type of adaptive immunity that is mediated by antibodies produced by plasma cells. Humoral immunity is the main mechanism for defending against extracellular microbes and their toxins.
•Cell-mediated immunity is the type of adaptive immunity mediated by T lymphocytes; cell-mediated immunity is the main defense mechanism against microbes that survive within phagocytes (i.e., bacteria that causes Tuberculosis) or that infect the cytosol of non-phagocytic cells (i.e., viruses).
•Cytokines are secreted proteins that work as mediators of immune and inflammatory reactions. Cytokines provide a mechanism for cells of the immune system to “talk” to one another to coordinate a response.
“In the end, it’s not the years in your life that count. It’s the life in your years.” Abraham Lincoln
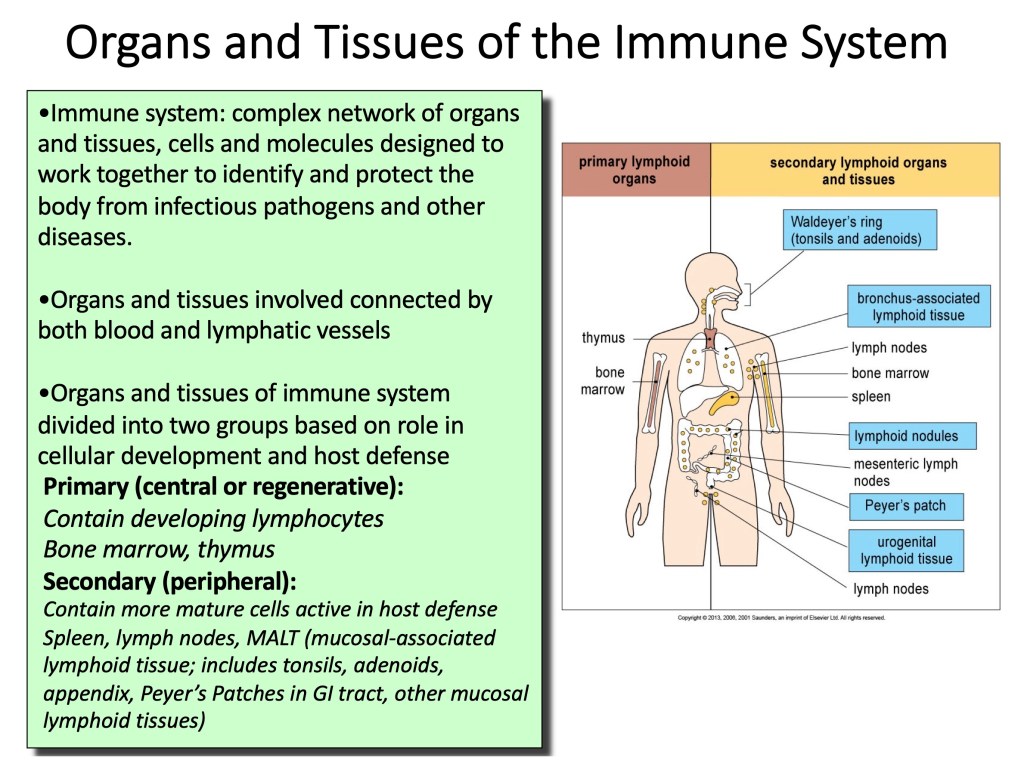
Organs and Tissues of The Immune System:
•Immune system: complex network of organs and tissues, cells and molecules designed to work together to identify and protect the body from infectious pathogens and other diseases.
•Organs and tissues connected by both blood and lymphatic vessels Organs and tissues of immune system divided into two groups based on role in cellular development and host defense.
“My health is good; it’s my age that’s bad.” Roy Acuff
Defining the Innate immune System:
•The battle against infection begins when resident macrophages eat the bacteria on the splinter in your finger;
•Substances generated increase flow of blood in wound;
•Other chemicals cause vessels to contract and fluids flow in;
•Macrophage compounds send pain signals to brain;
•Macrophages produce cytokines, which alert/recruit other immune system cells, and thus, the inflammatory response (battle) is on;
•Our innate immune system usually responds so quickly to common pathogens the battle may be over in just days.
•The innate immune system is a ‘hard-wired’ defense system that we are born with and never changes; consider the home telephone (hard-wired, unchanged) vs. the cell phone (mobile, constantly changing).
Differences Between Acute Inflammation versus Chronic Inflammation: We said this describing the importance of inflammation to the immune system, “An essential aspect of the immune system is inflammation, which is the immune system’s response to infection, injury, and toxic compounds. We consider the inflammatory process to be a host-defense response mechanism. Unfortunately, inflammation, if not controlled, can result in host morbidity and mortality.”
•Acute inflammation
–Immediate reaction to tissue injury
–Rapid onset
–Innate immunity
–Neutrophil cell predominance
–Duration hours to weeks
–Prominent vascular response
•Chronic inflammation
–Persistent reaction to tissue injury
–Slow response
–Cell-mediated immunity
–Mononuclear cell predominance (lymphocytes, plasma cells, monocytes/macrophages)
–Duration weeks to years
–Less prominent vascular response
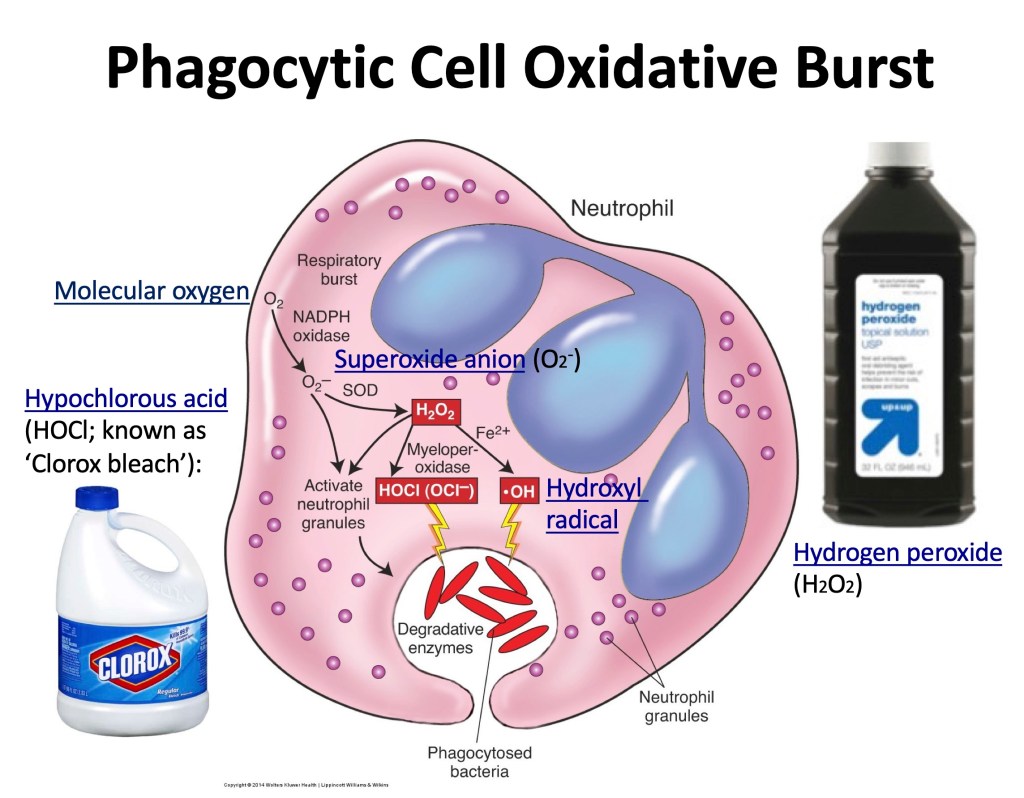
Phagocytic Cell Respiratory or Oxidative Burst: Order of Events:
1.Molecular oxygen reduced by NADPH oxidase produces superoxide anion (O2–):
NADPH Oxidase
2O2 + NADPH ——————> 2O2– + NADP+ + H+
2. Generation of hydrogen peroxide (H2O2) superoxide dismutase (SOD):
SOD
2O2– + 2H+ ——-> H2O2 + O2
Primary reactive species responsible for killing ingested microorganisms are generated from superoxide and hydrogen peroxide in a series of reactions:
3. Hydroxyl radical (OH•) attacks DNA
H2O2 + O2 ——————> OH• + OH- + O2
.4. Hypochlorous acid (HOCl) has many toxic effects.
myeloperoxidase
H2O2 + Cl– ———————> HOCl + H2O
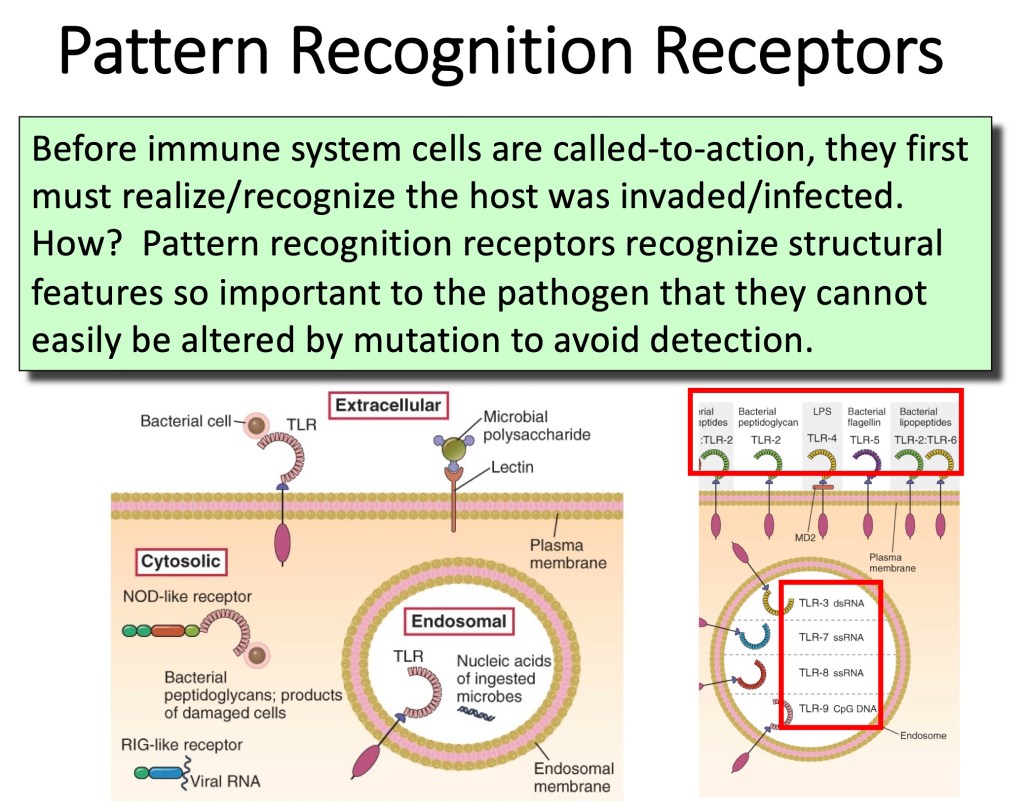
Pattern Recognition Receptors [Toll-like Receptors (TLR-1 through TLR-9)]:
The Type I Interferon System of Innate Immunity:
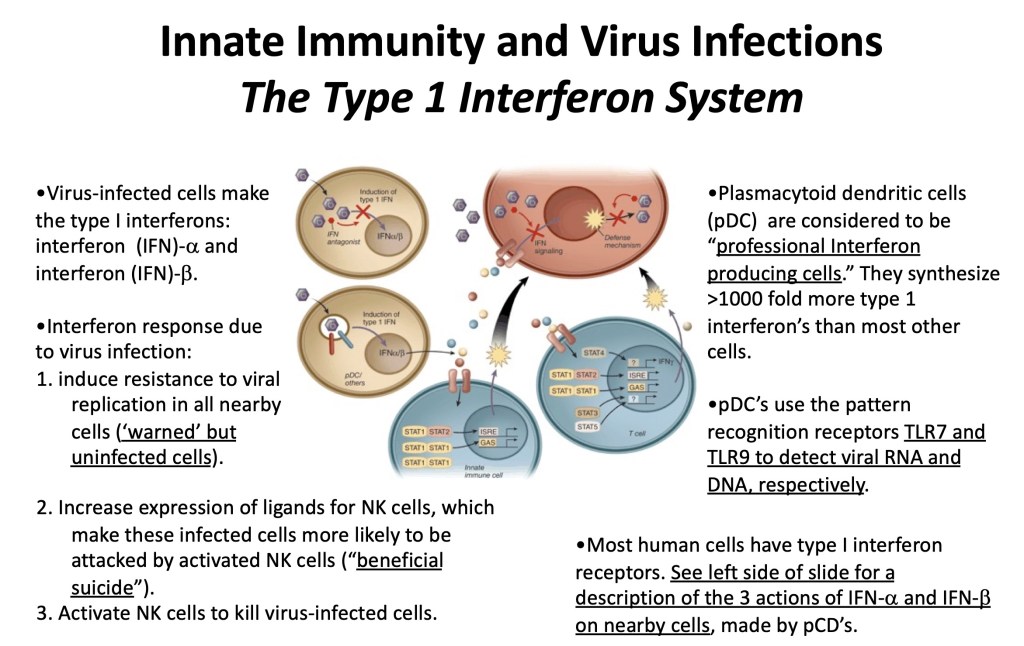
“You must become an old man in good time if you wish to be an old man long.” Marcus Aurelius
Defining Adaptive Immunity:
•Besides innate immunity, our host defense has adaptive immunity, which develops slowly but provides a more potent defense against infections.
•Adaptive immunity (also called acquired immunity) requires the expansion and differentiation of lymphocytes in response to pathogens before providing an effective defense.
•Together with the help of innate immunity but also the third line of defense against microorganisms;
•B lymphocytes – Secreted antibodies bind to extracellular microbes, which promotes their ingestion by phagocytic cells;
•Phagocytic cells ingest microbes, and helper T cells enhance the microbe-killing abilities of phagocytic cells;
•Helper T cells recruit leukocytes to destroy microbes and strengthen the epithelial barrier function to expel microbes;
•Cytotoxic T lymphocytes destroy cells infected by microbes that are inaccessible to antibodies.
Example of a Pro-Inflammatory Response is when Adaptive Immune Cells and Macrophages Encounter a Pathogen:
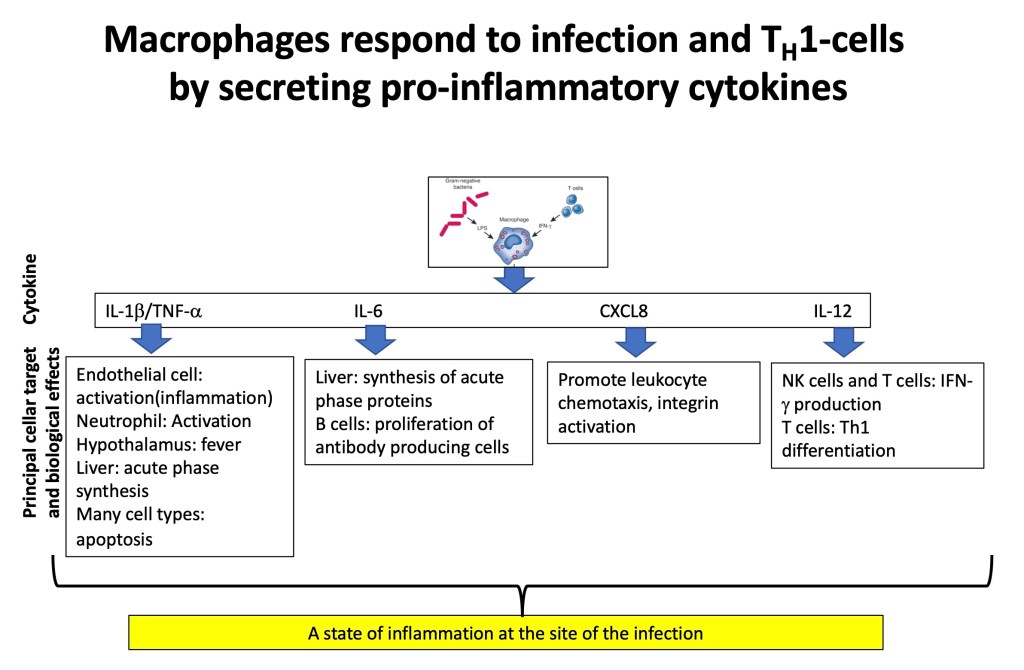
“I’m saving that rocker for the day when I feel as old as I really am.” Dwight D. Eisenhower
Topics NOT Included here in this Introductory Overview. As I said in the beginning, there is incredible richness and detail in the functioning of the immune system. My attempted goal has been to give one an appreciation of the immune system. Here is what will not be included because this blog post is just getting way too long:
The Complement System-Activity
Five functional Classes of Effector CD4 T cell are produced by activation and differentiation in different cytokine environments
Activation of Naïve T Cells
Presentation of Antigen by MHC to T Cells
Maturation of T Cells: “Thymic Education”
Co-stimulation is Required to Activate T Cells
B7/CD28 receptor ligand pairs vs. CD40/CD40L
Effector Functions of CD4+ Helper T Cells in Cell-mediated Immunity (CMI)
Effector Functions of CD4+ Helper T Cells in Humoral Immunity
Development and Function of TH1, TH2, and TH17 Effector Cells
CD8+ T Cells Differentiate into Cytotoxic T Cells or “CTL’s”
Properties of antibodies and T cell antigen receptors (TCRs)
Flow of Humoral Immunity
Functional Consequences of B Cell Activation by Antigen
Antibodies
B Cell Maturation and Selection
Antigen Receptor-mediated Signaling in B Cells
Primary and Secondary Antibody Responses
Function of Helper T Lymphocytes in Humoral Immune Responses to Protein Antigens
The germinal center reaction
Recombination and Expression of Immunoglobulin (Ig) Genes
Heavy-Chain Isotype (Class) Switching
Affinity Maturation of B Cells
Hypersensitivity and Autoimmunity
“When you are forty, half of you belongs to the past… And when you are seventy, nearly all of you.” Jean Anouilh
PART 2- Effect of Aging on the Immune System:
•One of the most recognized consequences of aging is a decline in immune function;
•Effects of aging include reduced production of B and T cells in the bone marrow and thymus and diminished function of mature lymphocytes in secondary lymphoid tissues;
•While older adult individuals are by no means immunodeficient, they often do not respond efficiently to novel or previously encountered antigens:
-illustrated by increased susceptibility of individuals >70 y.o. to influenza, a situation exacerbated by poor response to vaccination.
Hallmarks of Aging
•These hallmarks have been subdivided into primary, antagonistic, and integrative.
•The primary hallmarks, i.e., genomic instability, telomere attrition, epigenetic alterations, and loss of proteostasis, act as the instigators of cellular damage.
•The antagonistic hallmarks, deregulated nutrient sensing, mitochondrial dysfunction, and cellular senescence, are tasked with countering the damage inflicted on the organism but can compound it when exacerbated or persistent.
•The integrative hallmarks, stem cell exhaustion, and altered cellular communication emerge when the collective damage caused by the primary and antagonistic hallmarks cannot be reversed and are responsible for the physiological phenotype observed with age.
T Cell Development and Function in Older Adults:
•Several factors seem to contribute to T cell dysfunction in the elderly, among which thymic involution is a primary cause.
•Fewer new T cells emigrate from the thymus in adults and consequently, elderly individuals have fewer naïve T cells;
•There is an age-related over-representation of effector or memory T cells versus naïve T cells in the elderly;
•Naïve T cells in the elderly (70–85 years) are less functional in response to antigens than in young individuals ( 20–35 years).
Age-associated Changes in B cells:
•Antibody responses are impaired with aging, leading to problems such as reduced immune responses to vaccinations;
•Aging is associated with a dampening of B cell lymphopoiesis and attenuated CD4+ T cell help;
•Potent B cell-mediated antibody responses require help by CD4+ T cells, and findings show that aging causes defects in the function of CD4+ T cells upon antigen stimulation;
•B cells themselves become modulated with age. The B cell compartment and the antibody response’s quantity and quality are reduced in older adult individuals.
•A study of pneumococcal immunization in elderly versus young individuals showed age-associated losses of the protective high-affinity immune response;
•Older adult individuals have been shown to accumulate mutations in the immunoglobulin heavy chain genes in peripheral B cells;
•Other studies show an accumulation of gene mutations ultimately leading to B cell dysfunction in older individuals.
Age-related Changes in Innate Immune Cells:
•Advanced age has been associated with a change in not only the differentiation but also the phenotype of dendritic cells;
•Macrophages have reduced expression of major histocompatibility complex (MHC class II and other proteins, which ultimately has an impact on the function of these cells and their response to lipopolysaccharide;
•Aging also affects the co-stimulation of dendritic cells and impairs the function of neutrophils (decreased effector functions) and natural killer cells;
•Toll-like receptor (TLR) signaling pathways, particularly TLR7, TLR8, and TLR9 signaling, are altered in older adults.
Mechanisms of Immune Dysfunction with Aging:
•Both the adaptive and innate immune systems are altered as individual ages, becoming either hypo-functional or hyper-functional with time;
•A chronic low-grade inflammation occurs with age;
•Chronic inflammation is associated with the development of various chronic diseases;
•Cells of the innate and adaptive immune systems initiate and regulate inflammatory responses to pathogens and mediate chronic inflammation in aging.
Can Anything be Done to Combat the Eroding Immune System as One Ages: A few simple things come to mind, including taking vitamin D3 to boost the immune system (4,000-5,000 IU/day), moderate aerobic exercise keeps the pro-inflammatory: anti-inflammatory response better balanced, sleep more, reduce levels of stress in your life, maintain proper hygiene, make sure to have a heart- and brain- healthy diet, get pneumonia, influenza, and COVID-19 immunizations to better enable your immune system to react when necessary, and talk it over with your family physician.
“The secret of genius is to carry the spirit of the child into old age, which means never losing your enthusiasm.” Aldous Huxley
PART 3- Dysfunctional and Aging Immune System in the Development of Parkinson’s.
Neuroinflammation Drives the Process:
In describing the role of the immune system in developing Parkinson’s, we said, “When added to advanced age as the leading risk factor for developing Parkinson’s, the immune system’s prominent role, or lack thereof, becomes a factor in further understanding the etiology of the mid-brain degeneration of dopaminergic neurons seen in Parkinson’s.”
•The occurrence of intracellular inclusions named Lewy bodies is associated with the development of Parkinson’s.
•A significant component of Lewy bodies is aggregates of the protein alpha-synuclein (aSYN). Thus, aberrant aggregation of aSYN is likely a trigger for some or many of the biochemical changes within the dopaminergic neurons.
•A possible scenario leading to Parkinson’s in older adults includes aSYN aggregation in dopaminergic neurons.
•The response to aSYN aggregation is cytokine-driven neuroinflammation, enabling an age-linked immunologic dysfunction.
•There is also evidence that aSYN, usually an intracellular protein, becomes an autoantigen released into the extracellular neuronal cell spaces and then aggregates, further activating the immune response to engage the now deranged neuronal cells.
•Over time, neuronal cells become dysfunctional and continue to be engaged by the inflammatory and immunological system cells and substances; dopaminergic neurons are destroyed and slowly, Parkinson’s evolves over the years.
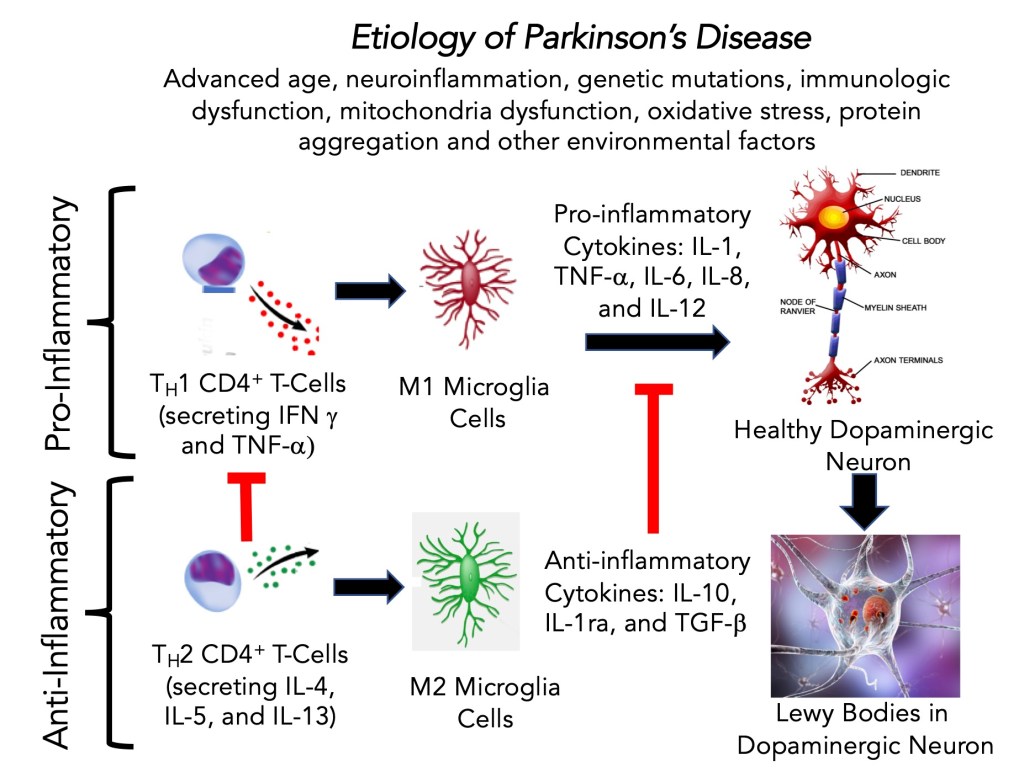
in the Central Nervous System. Sustained pro-inflammatory cytokines are linked to a shift in the balance of the immune system response to favor the pro-inflammatory state over the anti-inflammatory response. The pro-inflammatory state promotes the sustained activation and activity of M1 microglia cells (the macrophage-like cells in the brain).
“Age does not depend upon years, but upon temperament and health. Some men are born old, and some never grow so.” Tryon Edwards
Imbalance Between Pro-inflammatory and Anti-inflammatory White Blood Cells Contributes to the Development of Parkinson’s:
These steps are described in the drawing for the immune system’s potential role in the development of Parkinson’s.
•First, the pro-inflammatory state (neuroinflammation) prompted by the accumulating aSYN aggregates provides the first response.
•The response has TH1 CD4+ T cells supporting a pro-inflammatory response to activating M1 microglia cells.
•Second, in a typical immunological process, the anti-inflammatory arm of the system turns to TH2 CD4+ T cells and their cytokines activate anti-inflammatory M2 microglia cells that down-regulate the pro-inflammatory processes.
•This is the appropriate response to activate pro-inflammatory M1 microglia cells (the brain’s macrophages) that attempt to regulate the evolving problem, and then the appropriate anti-inflammatory response shuts it off.
•There is an imbalance in Parkinson’s to favor the pro-inflammatory CD4+ T cells, resulting in a sustained neuroinflammatory state.
•Combined with the known age-related dysfunction of the immune system described above, neuroinflammation does not abate, further enhancing a detrimental immunological response in the mid-brains of unknowing PD patients.
“Life can only be understood backwards.” Mackenzie Astin
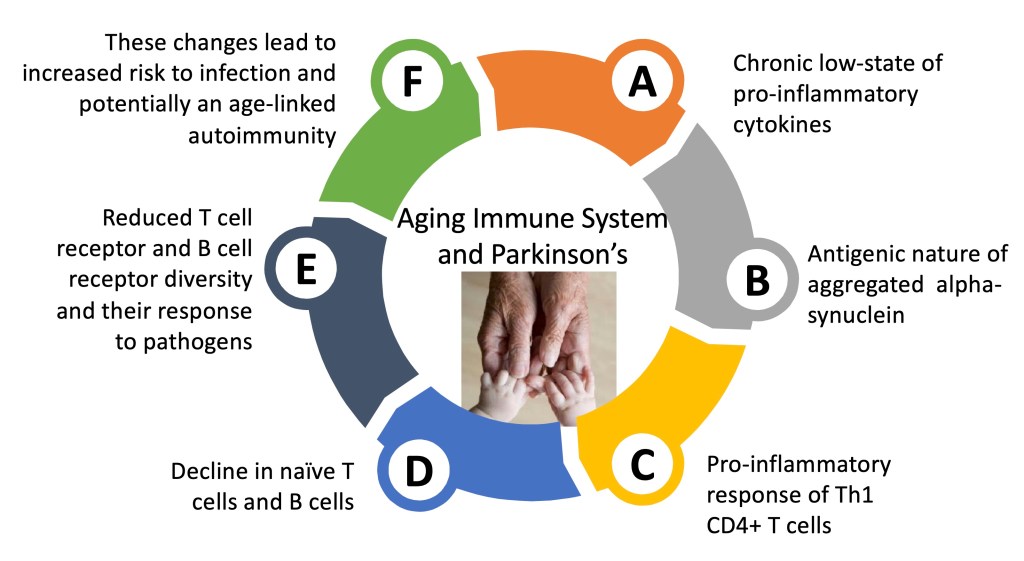
A Collection of Immunological Dysfunctions can be Summarized to Potentially Contribute to the Development of Parkinson’s: The schematic below represents some of the key immune dysfunctions that could potentially favor the development of Parkinson’s. This is likely not the only thing that contributes to Parkinson’s; however, the erosion of the immune system as we age likely partici[pates in the development of Parkinson’s.
“A man is not old until regrets take the place of dreams.” John Barrymore
The Future and Therapeutic Options: If you have gotten to the end of the blog post, there is substantial evidence to suggest the immune dysfunctions of an aging immune system can contribute to the development of Parkinson’s. Likely, however, there will need to be additional triggers that include viral and bacterial infections, environmental toxins, alterations in the gut microbiome, and other potential risk factors known to promote Parkinson’s.
In terms of therapy, more could be done to investigate the potential of anti-inflammatory agents in Parkinson’s, as is being done for other neurodegenerative disorders (Alzheimer’s disease and amyotrophic lateral sclerosis). Immune system markers as precursors could also be studied as the preface during the development of Parkinson’s. Newer generation anti-tumor necrosis factor compounds might help treat Parkinson’s as they penetrate the blood-brain barrier. Better treatment options would be helpful for the gut-microbiome axis to reduce the chronic inflammatory state. Finally, there is some older evidence that non-steroidal anti-inflammatory drugs (NSAIDs) reduced the risk of some neurodegenerative disorders, but this has not been as well-studied in Parkinson’s.
“I don’t know how I got over the hill without getting to the top.” Will Rogers
Useful References:
Tansey, M.G.; Wallings, R.L.; Houser, M.C.; Herrick, M.K.; Keating, C.E.; Joers, V. Inflammation and immune dysfunction in Parkinson disease. Nature Reviews Immunology, 2022, 1-17.
Hribar, C.A.; Cobbold, P.H.; Church, F.C. Potential role of vitamin D in the elderly to resist COVID-19 and to slow progression of Parkinson’s disease. Brain Sci. 2020, 10, 284. [Google Scholar]
Hall, M.-F.E.; Church, F.C. Exercise for older adults improves the quality of life in Parkinson’s disease and potentially enhances the immune response to COVID-19. Brain Sci. 2020, 10, 612. [Google Scholar]
Morowitz, J.M.; Pogson, K.B.; Roque, D.A.; Church, F.C. Role of SARS-CoV-2 in Modifying Neurodegenerative Processes in Parkinson’s Disease: A Narrative Review. Brain Sci. 2022, 12, 536. https://doi.org/10.3390/brainsci12050536
Odoj, K.; Brawek, B.; Asavapanumas, N.; Mojtahedi, N.; Heneka, M.T.; Garaschuk, O. In vivo mechanisms of cortical network dysfunction induced by systemic inflammation. Brain Behav. Immun. 2021, 96, 113–126. [Google Scholar]
Harms, A.S.; Ferreira, S.A.; Romero-Ramos, M. Periphery and brain, innate and adaptive immunity in Parkinson’s disease. Acta Neuropathol. 2021, 141, 527–545. [Google Scholar]
Mayne, K.; White, J.A.; McMurran, C.E.; Rivera, F.J.; de la Fuente, A.G. Aging and neurodegenerative disease: Is the adaptive immune system a friend or foe? Front. Aging Neurosci. 2020, 12, 305. [Google Scholar]
De Virgilio, A.; Greco, A.; Fabbrini, G.; Inghilleri, M.; Rizzo, M.I.; Gallo, A.; Conte, M.; Rosato, C.; Appiani, M.C.; De Vincentiis, M. Parkinson’s disease: Autoimmunity and neuroinflammation. Autoimmun. Rev. 2016, 15, 1005–1011. [Google Scholar]
Garretti, F.; Agalliu, D.; Lindestam Arlehamn, C.S.; Sette, A.; Sulzer, D. Autoimmunity in Parkinson’s Disease: The role of α-synuclein-specific T cells. Front. Immunol. 2019, 10, 303. [Google Scholar]
Sulzer, D.; Alcalay, R.N.; Garretti, F.; Cote, L.; Kanter, E.; Agin-Liebes, J.; Liong, C.; McMurtrey, C.; Hildebrand, W.H.; Mao, X. T cells from patients with Parkinson’s disease recognize α-synuclein peptides. Nature 2017, 546, 656–661. [Google Scholar]
Zhang, W.; Wang, T.; Pei, Z.; Miller, D.S.; Wu, X.; Block, M.L.; Wilson, B.; Zhang, W.; Zhou, Y.; Hong, J.-S. Aggregated α-synuclein activates microglia: A process leading to disease progression in Parkinson’s disease. FASEB J. 2005, 19, 533–542. [Google Scholar]
Klegeris, A.; Pelech, S.; Giasson, B.I.; Maguire, J.; Zhang, H.; McGeer, E.G.; McGeer, P.L. α-Synuclein activates stress signaling protein kinases in THP-1 cells and microglia. Neurobiol. Aging 2008, 29, 739–752. [Google Scholar]
Su, X.; Maguire-Zeiss, K.A.; Giuliano, R.; Prifti, L.; Venkatesh, K.; Federoff, H.J. Synuclein activates microglia in a model of Parkinson’s disease. Neurobiol. Aging 2008, 29, 1690–1701. [Google Scholar]
Lee, H.-J.; Patel, S.; Lee, S.-J. Intravesicular localization and exocytosis of α-synuclein and its aggregates. J. Neurosci. 2005, 25, 6016–6024. [Google Scholar]
“He who would pass his declining years with honor and comfort, should, when young, consider that he may one day become old, and remember when he is old, that he has once been young.” Joseph Addison
Cover photo credit by Larisa Koshkina from Pixabay