“No person was ever honored for what he received. Honor has been the reward for what he gave.” Calvin Coolidge
“The most reliable way to predict the future is to create it.” Abraham Lincoln
Introduction: Recently, Jeff Keegan asked me if I had seen the paper by Gil-Martínez et al., an established Parkinson’s research group in Spain (click here to read it)? Jeff also asked if this paper would alter the CAM strategy for treating my Parkinson’s (Complementary and Alternative Medicine (CAM) and Over-the-Counter Therapies in Parkinson’s)? This work was highlighted by the incomparable Simon Stott in the monthly Research Review- December 2018 of the ScienceofParkinsons.com blog (click here to read it). I read the paper with great interest, as I had written blog posts on the antioxidants, N-acetyl-cysteine [(NAC) click here to read it] and glutathione (click here to read it). The goal here is to summarize this research (this is open access, click here to download), describe mouse models of human diseases, compare mouse models of Parkinson’s to human Parkinson’s, present a brief overview of the physiology of aging, describe the drugs used here and how they relate to many complementary and alternative medicine (CAM) approaches in treating Parkinson’s.

“A people that value its privileges above its principles soon loses both.” Dwight D. Eisenhower
Of Mice and Men, and Human Diseases: I have spent the majority of my academic career studying serine proteases and serine protease inhibitors (serpins) and their role in two human diseases, venous thrombosis (blood coagulation) and breast cancer (cancer cell migration); only recently have I started exploring Parkinson’s. My laboratory group was supported by grants/fellowships from the National Institutes of Health [(NIH) National Heart, Lung, and Blood Institute (NHLBI), National Institute on Aging (NIA), National Institute of Neurological Disorders and Stroke (NINDS), and National Cancer Institute (NCI)], the American Heart Association, and the Susan G. Komen for the Cure.
We focused on three types of experiments. (A) In vitro experiments, which are performed in a laboratory setting outside a living organism [specifically, we did protein biochemistry, molecular biology, site-directed mutagenesis, computer-assisted molecular modeling, and cell culture of primary and immortalized cells]. (B) Ex vivo studies, which describe experiments with tissue from an organism in an external environment with minimal alteration of natural conditions [we did immunohistochemistry of human tissue (normal and diseased), 3D-cell culture, and cell-cell co-culture]. And (C) in vivo model systems, which are experiments performed in a living organism [mouse models of hemostasis-thrombosis]. We achieved numerous goals and had meaningful publications from our in vitro and ex vivo studies. A highlight of an in vivo study was by Cardenas and others (click here to view it). We described an essential link to aging and venous thrombosis. Notably, we showed that the biological process called senescence contributed to the disease process of venous thrombosis. We then developed additional mouse models and studied venous thrombosis as it related to aging mice. This resulted in another paper (click here to view it), with a very different conclusion. Here, Cardenas et al. noted, “Our data suggest that natural aging in mice does not accurately represent the age-related hyper-coagulability and increase in venous thrombosis seen in humans.” Thus, our studies describe the difficulty in using a mouse to mimic human diseases.


“We can complain because rose bushes have thorns, or rejoice because thorn bushes have roses.” Abraham Lincoln
Comparing Mouse Models to Human Diseases: The experimental condition by model systems in mice assumes this will translate to the comparable human situation. Both Perlman and Seok et al. review how difficult it is to recapitulate the human condition in a mouse. They reveal that the mouse is not a perfect mimic for some human diseases; and as with all experiments, it is just one means to test a scientific hypothesis. Importantly, both Seok et al. and Perlman highlight the importance of mouse studies for our understanding of human biology. Seok et al. noted (click here to view paper), “A cornerstone of modern biomedical research is the use of mouse models to explore basic pathophysiological mechanisms, evaluate new therapeutic approaches, and make go or no-go decisions to carry new drug candidates forward into clinical trials.” Furthermore, Perlman stated (click here to view paper), “The use of mice as model organisms to study human biology is predicated on the genetic and physiological similarities between the species.” They conclude the mouse will continue to be the most useful model system for human diseases, but it is far from perfect at mimicking some human diseases.


“For this is what America is all about. It is the uncrossed desert and the unclimbed ridge. It is the star that is not reached and the harvest that is sleeping in the unplowed ground.” Lyndon B. Johnson
Of Mice and Men, and Parkinson’s Disease: Mouse models have been significant in furthering our understanding of Parkinson’s. I am convinced many tremendous advances have been achieved and are currently being done with the mouse and Parkinson’s. If you want to know the details, here are some excellent review articles that highlight the various mouse models of Parkinson’s (some are shown here and others are given at the end). Using different chemical reagents injected into the mouse, they develop a Parkinsonian-like syndrome. And the newer genetic models show some of the signs and symptoms of Parkinson’s. However, as outlined above, these model mouse systems are not perfect in recapitulating the human condition; yet, they offer substantial scientific knowledge of Parkinson’s if interpreted with some caution.






“In matters of style, swim with the current; in matters of principle, stand like a rock.” Thomas Jefferson
•The “Tool Box” for Gil-Martínez et al. “Unexpected Exacerbation of Neuroinflammatory Response After a Combined Therapy in Old Parkinsonian Mice” (2018):
•They used aged mice (20 months old mice are ~65 years old in human years) with the MPTP-toxin model of Parkinson’s.
•The goal was to determine the potential neuroprotective role from combining the anti-oxidant N-acetyl cysteine (NAC) with the anti-inflammatory HA-1077 (also known as Fasudil).
•Unexpectedly, the combined use of NAC (100 mg/Kg i.p.) and HA-1077 (40 mg/Kg i.p.) produced a huge increase in dopaminergic neuronal cell death in the MPTP-model compared to control mice.
•NAC and HA-1077 together led to both microglial and astroglial cell activation in the midbrain region of MPTP-treated mice.
•Interestingly, when used alone, NAC was very effective at preserving neuronal cell death in the MPTP-model of Parkinson’s.
•Correlates were made to aging humans with Parkinson’s and using a combination of drugs that could potentially and unexpectantly be detrimental.
“Believe you can and you’re halfway there.” Theodore Roosevelt
Brief Overview of the Aging Adult: An important feature of their research was using an aged mouse of 20 months, which corresponds to ~65 years old in human years. The physiology of aging is complex and will be summarized in a future blog post. However, here is a preview.
Theories of Aging- The”Wear and Tear” theory states that cells are damaged by toxins from various sources, and the process to repair these cells-tissues is lost over time. “Antagonistic Pleiotropy” is the application of evolutionary mechanisms to aging. Basically, genes are pleiotropic; that means they control more than one trait or property. For example, genes that solidify chances of reproductive success but may be deleterious in later life are still selected. Thus, aging is an outcome of evolutionary selection. The “Free Radical” theory states that repeated and progressive reactive oxygen species (ROS) oxidatively damage cells and tissues, and there is less ability with advanced age to repair or regenerate cells/tissues. Regardless of which theory of aging is most correct, the mechanism to repair the damage to cells/tissues is dysfunctional with respect to advanced aging.
Changes in the Brain with Aging- Aging produces a decrease in neural density. Central neurotransmitters are reduced, including catecholamines, serotonin, and acetylcholine. These changes alter mood, memory and motor function. There is an age-related deficiency of dopamine uptake sites and transporters, with additional loss of alpha-2-adrenergic, beta-adrenergic and gamma-aminobutyric acid binding sites. These alterations lead to a reduction in the speed of processing and memory.
Adverse Drug Reaction in Advanced Age- Adverse drug reactions increase with the number of drugs administered; this adverse drug reaction happens linearly as a function of age. Alterations in renal, CV, hepatic function may change drug concentrations and their duration of action. The decrease in lean body mass with an increased proportion of body fat. As mentioned recently here (Managing Chronic Pain in Parkinson’s Disease), the American Geriatric Society annually publishes a guide called the Beers Criteria. The Beers Criteria is an important document that lists the medications to be avoided in older adults.
“You can’t let your failures define you. You have to let your failures teach you.” Barack Obama
Mechanism of Anti-inflammatory HA-1077 (Fasudil) Compared to Other Anti-inflammatory Substances: The anti-inflammatory studied here was Fasudil (or HA-1077). Fasudil hydrochloride (Eril®) is a selective RhoA/Rho kinase (ROCK) inhibitor. ROCK is an enzyme that plays an important role in mediating vasoconstriction and vascular remodeling in the pathogenesis of pulmonary hypertension. ROCK induces vasoconstriction by phosphorylating the myosin-binding subunit of myosin light chain (MLC) phosphatase, thus decreasing MLC phosphatase activity, and enhancing vascular smooth muscle contraction. Eril® is indicated for the treatment of cerebral vasospasm and cerebral ischemia. Eril® is available as an injection for intravenous use, containing 30 mg of Fasudil hydrochloride. The recommended dosage is 30 mg fasudil hydrochloride, diluted in 50-100 mL solution, and administered by intravenous infusion over 30 minutes 2-3 times daily.
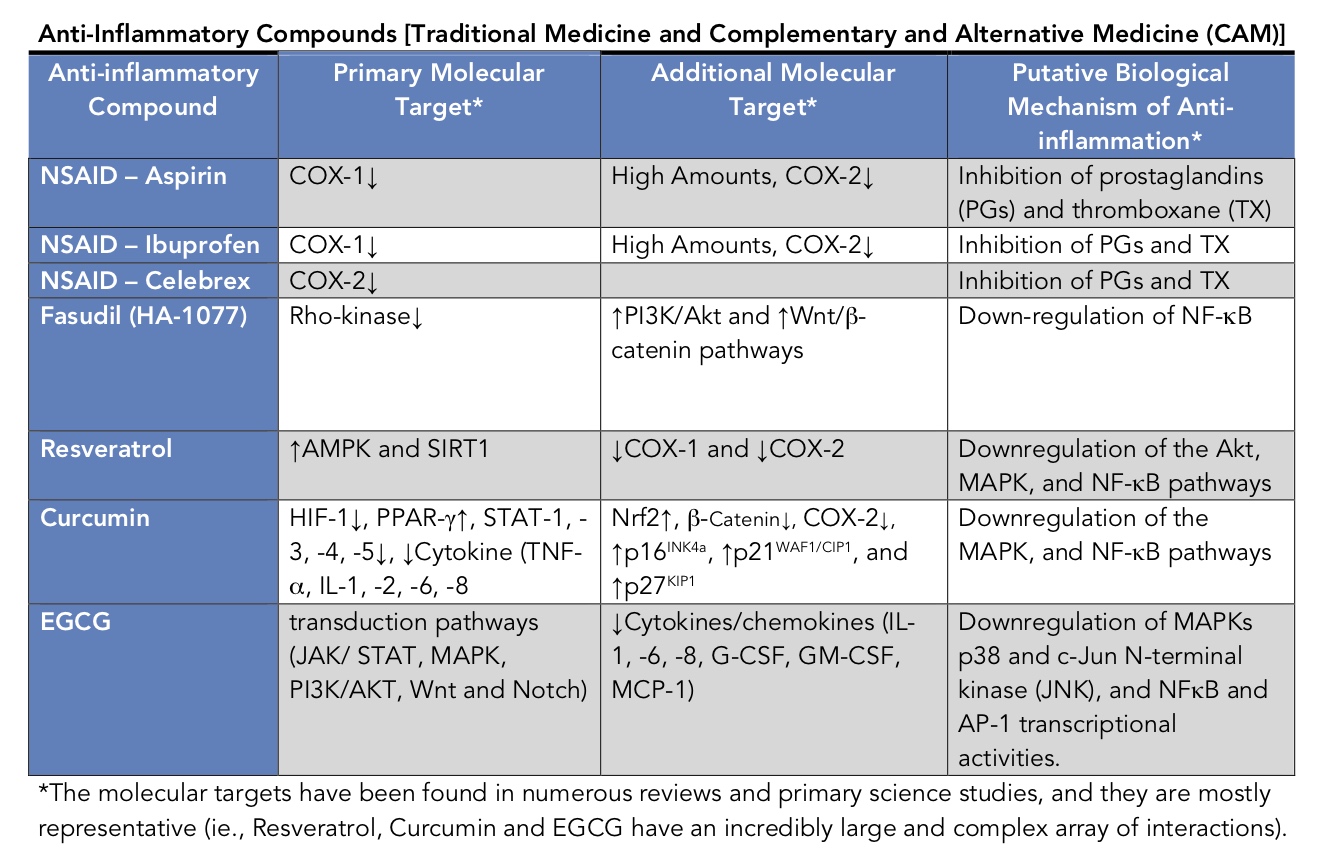
I was not familiar with Fasudil; thus, I made the table below comparing some anti-inflammatory compounds. Besides Fasudil, I have included compounds from two classes of anti-inflammatory drugs. First, from Traditional Medicine, there are three NSAIDs (Aspirin, Ibuprofen, and Celebrex). Next, there are three Complementary and Alternative (or CAM) substances (Resveratrol, Curcumin, and EGCG). An inspection of the Table below shows a large number of molecular targets; the most apparent molecule downregulated by many of the substances is NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells). NF-κB is a protein complex that is the master regulator of the transcription of DNA, cytokine production, and cell survival. Amongst the many different molecular targets, one result that stood out was that Fasudil activated PI3K/Akt while the CAM drugs inactivated related signaling substances. Therefore, a question we are left with is would the CAM substances and NSAIDs, as did Fasudil, when mixed with NAC create further neuronal cell death in an aged Parkinson’s mouse model or an aging human?
“One person can make a difference, and everyone should try.” John F. Kennedy
A “Cautionary Tail” (okay a Cautionary Tale) from an old Mouse with Parkinson’s: To recap, in a well-written and scientifically well-executed study, Gil-Martínez et al. showed that combining Fasudil (HA-1077, anti-inflammatory) with NAC (antioxidant) led to substantial dopaminergic neuronal cell death using a model of old Parkinson’s mice. Recently, Collier et al. studied the link between aging and Parkinson’s disease. Interestingly, they suggested age-related changes in the dopamine system are similar to what they called a vulnerable pre-parkinsonian state. The results from Gil-Martínez et al. imply we have increased susceptibility to Parkinson’s as we age due to some exaggerated response to combined drug therapy. This sinister synergy between NAC and HA-1077 (Fasudil) resulted in no neuronal cell resurrection, which was their original hypothesis.
Let’s relate drug doses used in the mouse and then compare it to a human. Experimentally, they used NAC (100 mg/Kg mouse weight i.p.) and HA-1077 (40 mg/Kg mouse weight i.p.). If we use 80 Kg = 176 lbs weight of a human (as an example), this would translate into using 8,000 mg of NAC (100 mg/Kg x 80 kg weight = 8,000 mg NAC being injected) and 3,200 mg of HA-1077 [Fasudil (40 mg/Kg x 80 Kg)], respectively. The typical daily dose of NAC is usually 1800 mg (3 x 600 mg capsule orally). The recommended dose of Fasudil is 30 mg x 2-3 times/day = 60-90 mg of Fasudil. Under these conditions, it works out to be a 4.44 fold-excess of NAC experimental dose/typical dose and 35.5-53.3 fold-excess of Fasudil (HA-1077) experimental dose/recommended dose.
What does this say about someone who combines antioxidant and anti-inflammatory CAM components in an attempt either to possibly provide neuroprotection or to slow progression of the disease? And let’s say this person is 65 years of age or older. If there is a strong correlation with a Parkinson’s mouse to human, this definitely sends up a red warning flag (the warning flags described here are part of the eight flags used in NASCAR racing; click here for further information). We will raise a black warning flag if the Parkinson’s mouse to human relationship is weak. Of course, drug dosage would likely be significant; the dosages of both NAC (the antioxidant) and HA-1077 (Fasudil the anti-inflammatory) were substantially higher than usual recommended dosages for each substance. Furthermore, the target specificity of both the CAM substances (Resveratrol, Curcumin, and EGCG) and the NSAIDs (Aspirin, Ibuprofen, and Celebrex) are seemingly very different to HA-1077 (although the CAM compounds and HA-1077 both down-regulate NF-kB, the paths used to get there are different).
The results from Gil-Martínez et al. were definitely surprising. Does this old Parkinson’s mouse model really relate to an aging human with Parkinson’s? It brings back a point made above. As mentioned here (Managing Chronic Pain in Parkinson’s Disease), older humans do have adverse drug interactions that do not occur in younger ages. It is not unreasonable to construct a comprehensive list of all drugs we are taking (for Parkinson’s and for other disorders) including traditional and CAM substances (by prescription and over-the-counter), and re-visit our Neurologist and/or Primary Care Physician for a consultation. Could some combination of conventional drugs we are currently taking actually be detrimental by enhancing neurodegeneration? Could some mixture of CAM compounds be in a threatening situation that promotes neuronal cell destruction, not the hoped-for neuroprotection? The yellow warning flag is being raised, which means to proceed with caution.
Please remember the material provided here is for educational and scientific purposes only. Under no circumstance should you consider this blog post as medical advice. Many of the substances described here are clinically available and some are even available over-the-counter; however, they may have serious side effects. Please communicate with your Neurologist or family-based Physician before any attempt to alter a treatment regime. “JourneywithParkinsons.com” cannot be held responsible for any actions taken based on the information provided here.
“Nothing in the world can take the place of persistence. Talent will not; nothing is more common than unsuccessful men with talent. Genius will not; unrewarded genius is almost a proverb. Education will not; the world is full of educated derelicts. Persistence and determination alone are omnipotent.” Calvin Coolidge
Literature Cited:
Bedell, M. A., N. A. Jenkins and N. G. Copeland (1997). “Mouse models of human disease. Part I: techniques and resources for genetic analysis in mice.” Genes & development 11(1): 1-10.
Bedell, M. A., D. A. Largaespada, N. A. Jenkins and N. G. Copeland (1997). “Mouse models of human disease. Part II: recent progress and future directions.” Genes & development 11(1): 11-43.
Bobela, W., L. Zheng and B. L. Schneider (2014). “Overview of mouse models of Parkinson’s disease.” Current protocols in mouse biology 4(3): 121-139.
Bove, J., D. Prou, C. Perier and S. Przedborski (2005). “Toxin-induced models of Parkinson’s disease.” NeuroRx 2.
Cardenas, J., M. Aleman, J. G. Wang, H. Whinna, A. Wolberg and F. Church (2013). “Murine models do not recapitulate the pathophysiology of age‐related venous thrombosis in humans.” Journal of Thrombosis and Haemostasis 11(5): 990-992.
Cardenas, J. C., A. P. Owens III, J. Krishnamurthy, N. E. Sharpless, H. C. Whinna and F. C. Church (2011). “Overexpression of the cell cycle inhibitor p16INK4a promotes a prothrombotic phenotype following vascular injury in mice.” Arteriosclerosis, thrombosis, and vascular biology 31(4): 827-833.
Cashman, J. N. (1996). “The mechanisms of action of NSAIDs in analgesia.” Drugs 52(5): 13-23.
Collier, T. J., N. M. Kanaan and J. H. Kordower (2017). “Aging and Parkinson’s disease: different sides of the same coin?” Movement Disorders 32(7): 983-990.
Dauer, W. and S. Przedborski (2003). “Parkinson’s disease: mechanisms and models.” Neuron 39.
Gerlach, M. and P. Riederer (1996). “Animal models of Parkinson’s disease: an empirical comparison with the phenomenology of the disease in man.” Journal of neural transmission 103(8-9): 987-1041.
Gil-Martínez, A. L., L. Cuenca, C. Estrada, C. Sánchez-Rodrigo, E. Fernández-Villalba and M. T. Herrero (2018). “Unexpected Exacerbation of Neuroinflammatory Response After a Combined Therapy in Old Parkinsonian Mice.” Frontiers in Cellular Neuroscience 12(451).
Gubellini, P. and P. Kachidian (2015). “Animal models of Parkinson’s disease: an updated overview.” Rev Neurol 171.
Hashimoto, M., E. Rockenstein and E. Masliah (2003). “Transgenic models of alpha-synuclein pathology: past, present, and future.” Ann N Y Acad Sci 991.
Jackson-Lewis, V., J. Blesa and S. Przedborski (2012). “Animal models of Parkinson’s disease.” Parkinsonism & related disorders 18: S183-S185.
Kulkarni, S. S. and C. Cantó (2015). “The molecular targets of resveratrol.” Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1852(6): 1114-1123.
Meredith, G. E. and D. J. Rademacher (2011). “MPTP mouse models of Parkinson’s disease: an update.” J Park Dis 1.
Miller, R. M., G. L. Kiser, T. Kaysser-Kranich, C. Casaceli, E. Colla and M. K. Lee (2007). “Wild-type and mutant alpha-synuclein induce a multi-component gene expression profile consistent with shared pathophysiology in different transgenic mouse models of PD.” Exp Neurol 204.
Ohishi, T., S. Goto, P. Monira, M. Isemura and Y. Nakamura (2016). “Anti-inflammatory action of green tea.” Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Inflammatory and Anti-Allergy Agents) 15(2): 74-90.
Rizzati, V., O. Briand, H. Guillou and L. Gamet-Payrastre (2016). “Effects of pesticide mixtures in human and animal models: an update of the recent literature.” Chem Biol Interact 254.
Rosenthal, N. and S. Brown (2007). “The mouse ascending: perspectives for human-disease models.” Nature cell biology 9(9): 993.
Schmidt, W. J. and M. Alam (2006). “Controversies on new animal models of Parkinson’s disease pro and con: the rotenone model of Parkinson’s disease (PD).” J Neural Transm Suppl 70.
Tieu, K. (2011). “A guide to neurotoxic animal models of Parkinson’s disease.” Cold Spring Harbor perspectives in medicine 1(1): a009316.
Vingill, S., N. Connor-Robson and R. Wade-Martins (2018). “Are rodent models of Parkinson’s disease behaving as they should?” Behavioural brain research 352: 133-141.
Zhou, H., C. S Beevers and S. Huang (2011). “The targets of curcumin.” Current drug targets 12(3): 332-347.
Cover Photo Credit: https://weheartit.com/entry/37489392


I’m so glad my question(s) inspired you to write this blog post. Now, if I could just get Simon to write a blog post about your blog post 🙂
LikeLike
Thanks for inspiring me. It took a while to write it. So why do you want Simon to write a blog about the same topic?
LikeLike
Yes, I realised after I posted it, that my joke was not very clear. The joke was about me being lazy, and hoping that Simon would simplify your blog post for me, thus saving me from reading your blog post several times (as I usually do). My background is not in chemistry, biology, or anything related, but I can usually get a fair understanding of your blog posts (and Simon’s too) by reading them several times.
Once I’ve done that, I may have a sensible question to ask!
LikeLike