“To find joy in work is to discover the fountain of youth.” Pearl S. Buck
“In dire times you can lose joy, but you can’t lose hope. Hope is your guide.” Paulo Coelho
Introduction: If you are living with Parkinson’s, you are living with some hope. You hope Michael J. Fox will one day soon announce, “Here is the cure for Parkinson’s!” Maybe you are hopeful that your newfound renewal of a rigorous exercise program will slow down the progression of your disorder. You are also hopeful that your next visit with your Neurologist will not require an increase in your anti-Parkinson’s therapy.
Furthermore, I will add one. I hope we find a better disease description that leads to an effective and novel treatment strategy for Parkinson’s. The story below about the somato-cognitive action network (SCAN) provides new insight into the pathogenesis of Parkinson’s disease and a potential non-invasive treatment. This is also a history lesson about the discovery of the motor cortex, and it is truly an outstanding science story.
“Hope and fear cannot occupy the same space. Invite one to stay.” Maya Angelou
The Paper: Recently, Ren et al. published a very interesting paper in Nature. Here is the citation for your further interest: Ren J, Zhang W, Dahmani L, Gordon EM, Li S, et al. Parkinson’s disease as a somato-cognitive action network disorder. Nature. 2026 Feb 4. doi: 10.1038/s41586-025-10059-1. Epub ahead of print. PMID: 41639440.

As a scientist, I have always looked at a paper from a submission, revision, and acceptance standpoint. By that, I mean: when the paper was submitted for publication and peer review, what the reviewers said and suggested, the time it took the authors to reply (usually by doing more experiments), and then the final decision. This paper took 14 months from submission to acceptance. However, the usual role of reviewers is to improve the story and provide stronger support for the hypothesis, not to defeat or deflate it. There is a tremendous amount of data presented here, and it is unbelievably precise and in detail.
In a brief side-step to illustrate the problem described above, we presented some interesting data on the relationship between aging and venous thrombosis. This was the culmination of work we had been developing for over four years [see Cardenas, Jessica C., A. Phillip Owens III, Janakiraman Krishnamurthy, Norman E. Sharpless, Herbert C. Whinna, and Frank C. Church. “Overexpression of the cell cycle inhibitor p16INK4a promotes a prothrombotic phenotype following vascular injury in mice.” Arteriosclerosis, thrombosis, and vascular biology 31, no. 4 (2011): 827-833].
The reviewers’ critiques were mostly positive, except that they wanted more data. They were asking us to prove it by doing a series of bone marrow transplants or crosses between the mice being studied. We could have submitted this work to a lesser journal; however, we embarked on a new clinical protocol, obtained new approval for animal procedures, performed a bone marrow transplant using irradiation, and, 10 months later, we had the answer. The revised manuscript was quickly accepted. Again, the reviewers were trying to improve the story; obtaining the answer was a major scientific challenge, but we succeeded in strengthening it.
Science is about presenting and explaining your hypothesis, answering the story with the best types of experiments possible, and then refining, retelling, and expanding upon what the data tells you about the original hypothesis. Thus, science is a hard task that demands attention to detail and involves developing and defending a scientific hypothesis.
“Hope is brightest when it dawns from fears.” Walter Scott
Discovery of the SCAN: As with most science, some observation, some idea, and some philosophy precedes a lot of scientific reports/observations. The story begins back in the 1930s, yes, many, many years ago. Wilder Penfield and Edwin Boldrey had studied more than 160 patients undergoing operations under local anesthetics; they applied electrical impulses to various regions of the patients’ cerebral cortex, and the patients reported whether they experienced a sensation or movement in a specific body part. According to their depictions, one-third of the motor and sensory cortical areas was dedicated to the hands, another third to the tongue (face, lips, and pharynx), and the final third to the rest of the body, including the trunk, legs, and arms. Here is the citation of the paper: Penfield, Wilder, and Edwin Boldrey. “Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation.” Brain, Volume 60, Issue 4, December 1937, Pages 389–443, https://doi.org/10.1093/brain/60.4.389

Move forward to 2023, and what was termed the motor cortex (M1) from Penfield and Boldrey has always been thought to consist of a continuous somatotopic homunculus that extended down from the precentral gyrus from foot to face [The precentral gyrus is a prominent ridge (gyrus) on the surface of the frontal lobe, located immediately anterior to the central sulcus (groove), which contains the primary motor cortex. A continuous somatotopic homunculus defines the traditional neuroanatomical map of the primary motor complex; this depicts an orderly, point-to-point map of the human body aligned in the cerebral cortex; and homunculus is Latin for “little man” or “little person.”].
Gordon et al., using precision functional magnetic resonance imaging (fMRI) methods, found something different from what was depicted in the “classical” report. They found that the “continuous” homunculus was interrupted by regions with distinct connectivity that alternated with effector-specific areas (foot, hand, and mouth). They found that these inter-effector regions had strong functional connectivity to the cingulo-opercular network (CON). The CON is necessary for action and physiological control, arousal, errors, and pain. Here is the citation for this paper: Gordon, Evan M., Roselyne J. Chauvin, Andrew N. Van, Aishwarya Rajesh, Ashley Nielsen, Dillan J. Newbold, Charles J. Lynch et al. “A somato-cognitive action network alternates with effector regions in motor cortex.” Nature 617, no. 7960 (2023): 351-359.

A major advance in the Gordon et al. paper was the redrawing of the M1 map by Penfield and Boldrey, shown below.
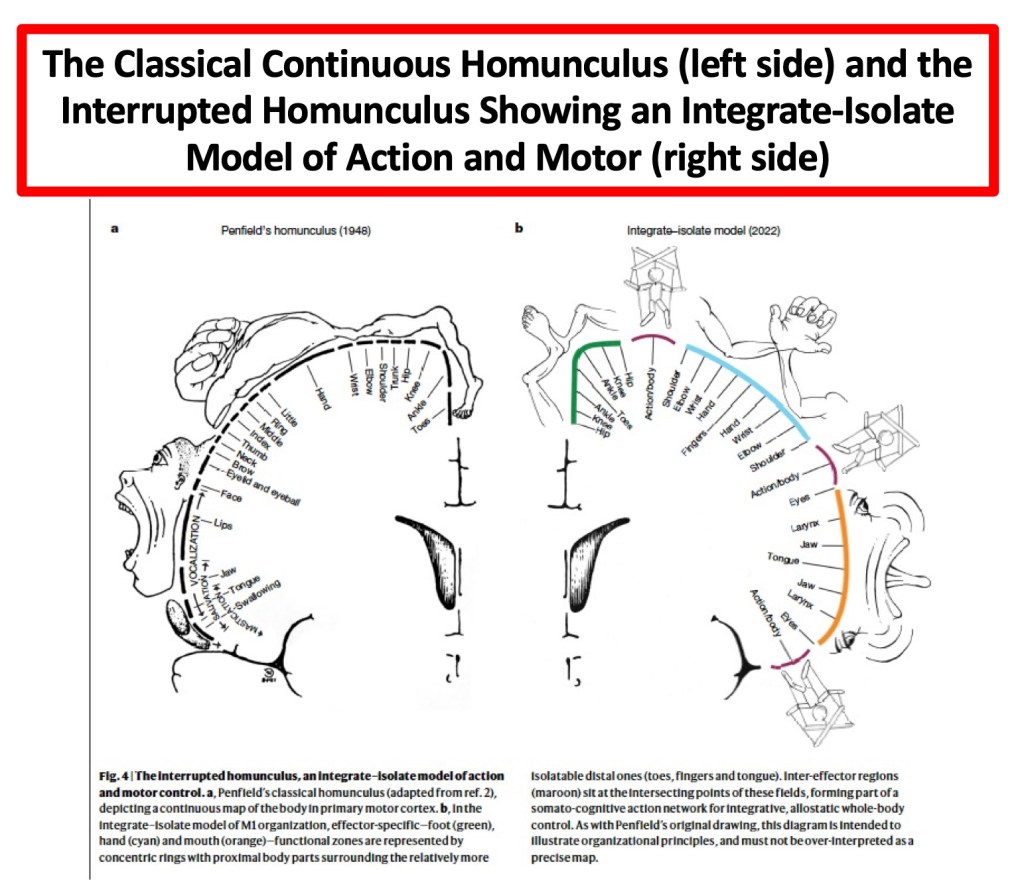
The interpretation of their data suggests that we have two behavioral control systems, contained and interwoven within human M1. The first is the well-described effector-specific circuits, which produce precise, isolated movements of our highly specialized appendages, such as our fingers, toes, and tongue. This describes the nimble motion we need for speaking and manipulating objects. The second unit is an integrative output system called SCAN (somato-cognitive action network), which controls the whole body. The SCAN integrates body control (motor and autonomic) and action planning, implying a higher level of executive control that requires movement coordination. The authors go further, saying that SCAN helps explain why mind and body states frequently interact (for instance, shoulder tension, increased heart rate, and “butterflies in the stomach”).
Finally, Gordon et al. describe that many of the symptoms of Parkinson’s are derived from motor, physiological, and intentional dysfunctions, which mirror SCAN connections linked to postural control, intention, and physiological regulation. This finally leads to the paper I began describing at the beginning of the blog post.
“It’s being here now that’s important. There’s no past and there’s no future. All there is ever, is the now.” George Harrison
The Problem Addressed by Ren and associates in the Nature Paper: Parkinsons is not a simple disease. No, Parkinson’s is confounded and complicated by motor and non-motor symptoms. If this were only related to our motor problems, what we know would make sense (or does it?). By that, the loss of dopaminergic neurons in the pars compacta of the susbtantia nigra, disrupts the cortico-basal ganglia-thalamic circuit that contains the subthalamic nucleus, ventral intermediate nucleus/centromedian nucleus of the thalamus, internal and external segments of the globus pallidus, putamen, and primary motor cortex. This circuit functions early in the disease process in response to levodopa. With time, we become less responsive to levodopa, and in advanced stagesof the disorder, we move to invasive and non-invasive treatment strategies. However, the discovery of SCAN may have altered the pathogenesis of Parkinson’s.
The SCAN regions, as they have been found to promote action implementation, axial movement, arousal, and autonomic control, collaborate with classical effector-specific motor regions of the central sulcus. The central sulcus is a groove in the cerebral cortex that separates the parietal and frontal lobes. It acts as a major boundary between motor and sensory functions.
Therefore, the authors hypothesized that since the action of Parkinson’s symptoms (action, motor, somatic, arousal, and cognitive) is not effector-specific, but they are multidomain and affect the entire body, then (a) SCAN dysfunction might contribute to the pathophysiology of Parkinson’s and (b) SCAN-specific neuromodulation might help treat the symptoms of Parkinson’s.
“We must accept finite disappointment, but never lose infinite hope.” Martin Luther King, Jr.
Evidence for SCAN’s Involvement in Parkinson’s: The research team analyzed brain imaging data from more than 800 participants across multiple institutions. They studied patients with Parkinson’s, some having received DBS surgery, patients with other movement disorders (essential tremor, dystonia, and ALS). They evaluated what they termed resting-state functional connectivity (RSFC), as was done in the above study.
The SCAN/effector (foot, hand, and mouth) elements exist in Parkinson’s- As described by Gordon et al., they found a distinctive SCAN pattern that alternates among foot, hand, and mouth effector units, illustrated below (including the nigrostriatal dopamine machinery).
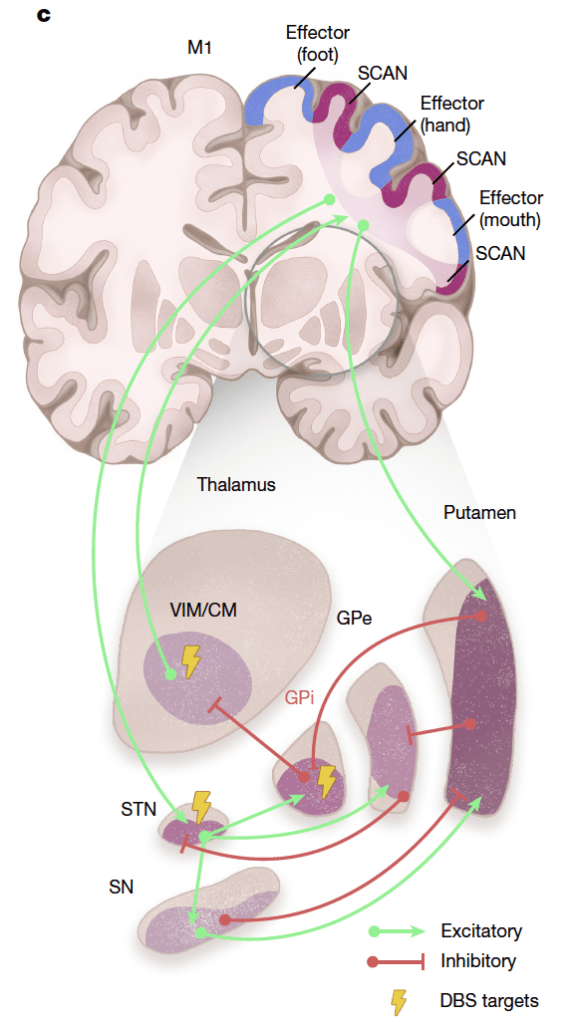
The dopamine machinery components preferentially interact with SCAN rather than with the effector elements. What they showed next was very interesting. The elements we know well in making dopamine, which are increasingly affected as Parkinson’s progresses, are more closely connected to the SCAN than to the effector-specific motor regions of the foot, hand, and mouth. The figure below illustrates this data using 226 scans from Parkinson’s patients, and look over the bar graph in the red box below. They compared how strongly connected each region of the subcortical structures, termed here as the cortico-basal ganglia-thalamic circuit [sustantia nigra (SN), subthalamic nucleus (STN), ventral intermediate nucleus/centromedian nucleus of the thalamus (VIM/CM), globus pallidus internus (GPi) and externus (GPe), and putamen], connected to the SCAN and the effector-specific units (foot, hand, and mouth). All six of the Parkinson’s-related circuit components interacted with significant statistical differences in the SCAN compared to the effector.
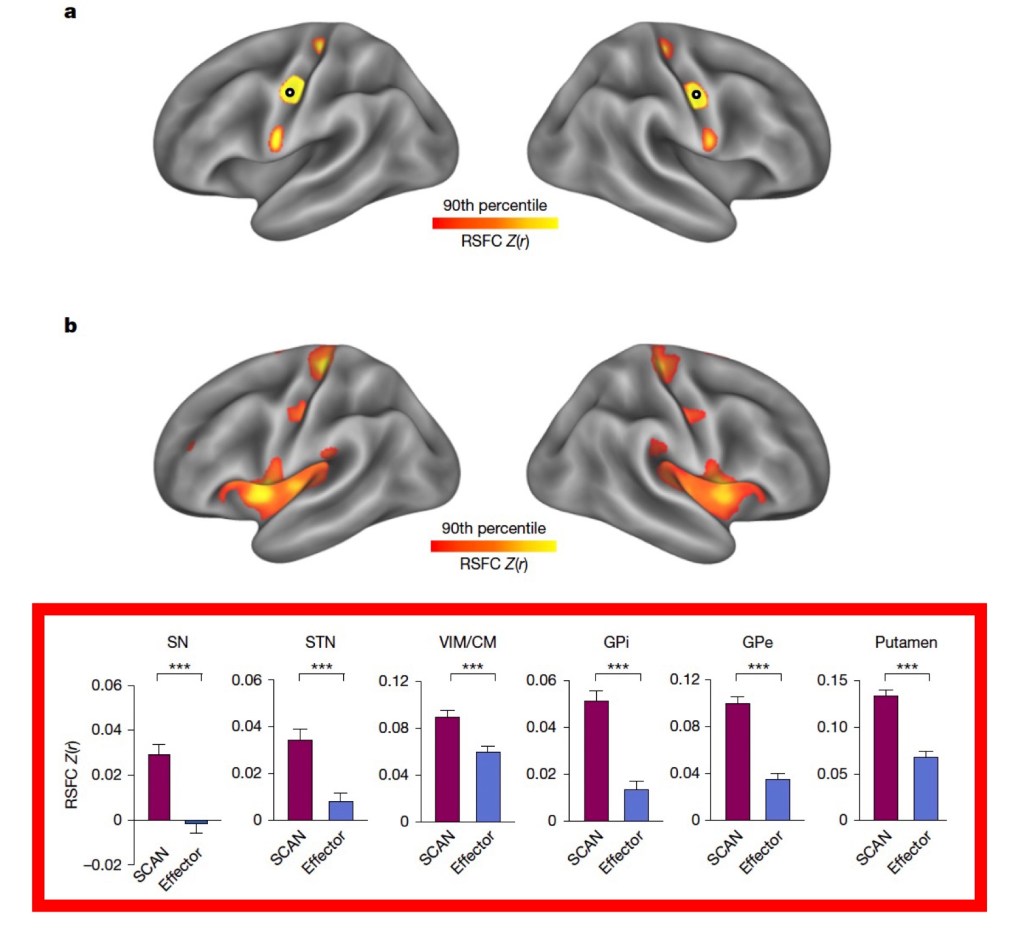
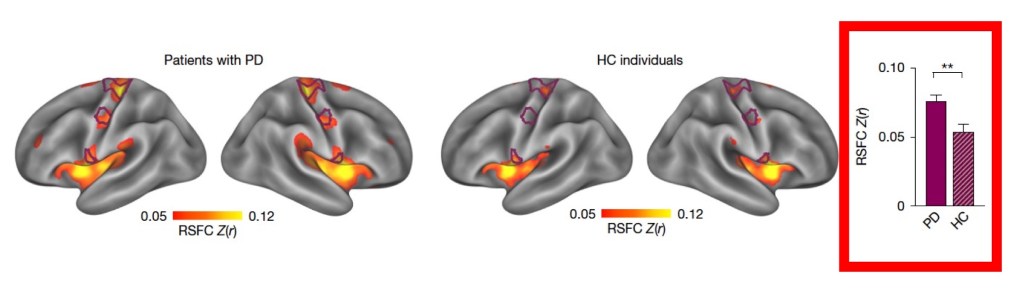
SCAN hyperconnectivity in Parkinson’s- They next asked whether the dopamine machinery components interacted with SCAN, comparing 65 Parkinson’s patients and aged-matched controls (60 individuals). The results are shown below. Looking again at the bar graph on the right side of the figure shows a significant increase in interaction between the dopamine elements and SCAN in Parkinson’s patients compared to the healthy control group. This suggests that there is a “hyperconnectivity” in Parkinson’s. They went further in their analysis and suggest that hyperconnectivity to SCAN was especially relevant for SCAN interactions with the substantia nigra, subthalamic nucleus, globus pallidus externus, and putamen.
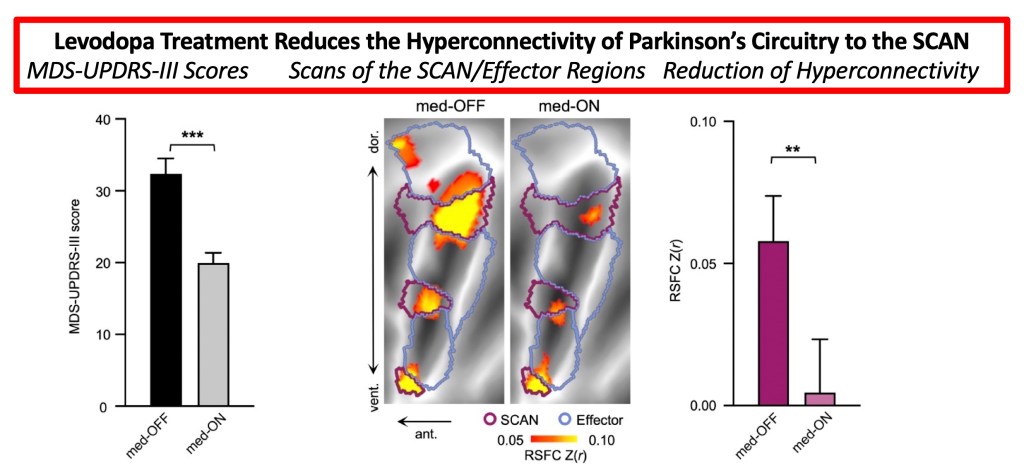
Levodopa treatment reduced SCAN hyperconnectivity. They showed that levodopa treatment was linked to improved UPDRS-III scores comparing ‘on’ to ‘off’ times, and there was a substantial reduction in hyperconnectivity to the SCAN in the ‘on’ phase compared to the ‘off’ phase. The figure below shows the change in MDS-UPDRS-III scores from levodopa treatment (left panel) and the change in hyperconnectivity to the SCAN (right panel). Consistent with the effect of levodopa altering the dopamine machinery by reducing SCAN hyperconnectivity is shown in the middle panel (the SCAN is located in the purple line areas, and the area shows a reduction in intensity going from ‘off’ to ‘on’ with levodopa).
Here is a summary of some additional studies (this is a long and detailed paper that goes much further in their analyses of Parkinson’s and SCAN then I will cover/review)-
•Subthalamic nucleus modulated by DBS surgery was found to be functionally connected to SCAN rather than effector-specific motor cortex regions.
•They then studied whether DBS normalizes the hyperconnectivity to SCAN in Parkinson’s. Motor symptoms improved at 1, 3, 6, and 12 months after DBS surgery (compared with pre-surgery MDS-UPDRS-III scores). The SCAN hyperconnectivity was lower at the 1-, 6-, 9-, and 12-month follow-up time points (the 3-month value was lower but not statistically significant). There was no change regarding the effector-specific units. This shows that the subthalamic nucleus, modulated by DBS surgery, reduces the hyperconnectivity of the Parkinson’s circuitry to the SCAN.
•They suggest that SCAN hyperconnectivity may serve as a biomarker for Parkinson’s. When studied in other movement disorders (essential tremor, dystonia, and ALS), SCAN hyperconnectivity was absent.
•The authors used the SCAN circuit to drive focused ultrasound thalamotomy. They found that lesions closer to the SCAN areas provided better clinical outcomes.
•Modulating the SCAN improved Parkinson’s treatment outcomes- They tested whether direct treatment of the SCAN could alleviate motor symptoms. They utilized a non-invasive form of repetitive transcranial magnetic stimulation (TMS). They targeted either the SCAN (18 Parkinson’s patients) or the effector-targeting group (18 Parkinson’s patients), and each group received 2 weeks of intermittent theta-burst stimulation. During and after the treatment procedures, both groups improved their MDS-UPDRS-III scores; however, the SCAN group had a greater reduction in scores. The SCAN group had reduced hyperconnectivity to the SCAN. Interestingly, the SCAN treatment group had significantly faster reductions in bradykinesia, rigidity, tremor, and axial symptoms. Although this is a small test group, these preliminary results suggest that a larger multicenter trial merits consideration.
“Totally without hope one cannot live. To live without hope is to cease to live. Hell is hopelessness. It is no accident that above the entrance to Dante’s hell is the inscription: “Leave behind all hope, you who enter here.”” Fyodor Dostoevsky
Conclusions: The paper by Ren et al. is the start of a very interesting and exciting idea: targeting SCAN and, by reducing hyperconnectivity between it and dopamine-producing components, could slow or even reduce the progression of the disorder. Would regular and vigorous exercise alter the SCAN hyperconnectivity? They suggest that cognitive and other non-motor symptoms may respond to altering the SCAN hyperconnectivity. That is just the start of the story. Of course, the causes of SCAN’s hyperconnectivity should be explored, as should many other questions.
The data provided in the paper and supplement files is substantial. I am sure others would have shown different aspects of their work- I tried to review experiments that I thought everyone who reads this blog would find helpful. I encourage you to read the paper and see if my “Cliff Notes” version of their paper is helpful.
Reading this type of paper gives me hope that one day, better treatment strategies will appear. My hope that science will step up and help us all is rooted in my 40-something years of science research and education. Parkinson’s is just waiting for the next set of scientists and clinicians to find an idea, explore the mystery, and provide useful answers that pave the way for a successful treatment. Ren et al. have opened a promising door to harnessing the hyperconnectivity of the SCAN region of the motor cortex to treat Parkinson’s disease symptoms and potentially slow disease progression. As Lao Tzu said, “There is a time to live and a time to die, but never to reject the moment. “
“Hope is to our spirits what oxygen is to our lungs. Lose hope and you die. They may not bury you for awhile, but without hope you are dead inside. The only way to face the future is to fly straight into it on the wings of hope….hope is the energy of the soul. Hope is the power of tomorrow.” Lewis B. Smedes