“Some remedies are worse than the disease.” Publilius Syrus
“Each patient carries his own doctor inside him.” Norman Cousins
Summary: Dopamine agonists are widely used in the treatment of Parkinson’s, especially as a first-line therapy. Some patients on a dopamine agonist experience side-effects that require either tapering or discontinuation of the drug. First described in 2010, dopamine agonist withdrawal syndrome (DAWS) is a complication of ~20% of Parkinson’s patients who are either lowering or stopping the dopamine agonist. DAWS presents as a cluster of physical and behavioral symptoms [e.g., agitation, depression, drug craving, and panic attacks (to give a few possible symptoms)]. There is no known standard-of-care in dealing with DAWS in Parkinson’s. Presented here is a brief overview of DAWS in Parkinson’s including dopamine agonists, clinical description, risk factors and prevalence, mechanism of action, treatment/management, and key publications.
“To heal illness, begin by restoring balance.” Caroline Myss
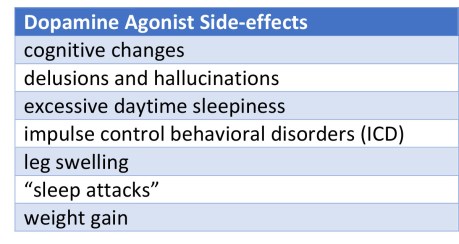
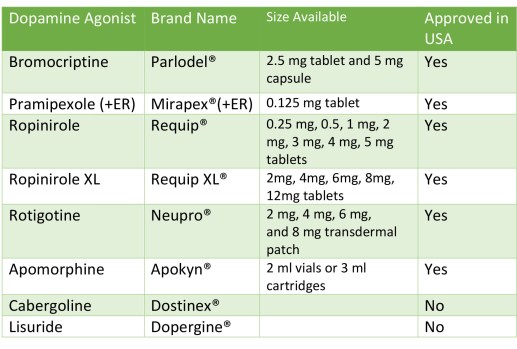
Dopamine agonists (DA): Dopamine agonists are ‘mimics’ of dopamine that pass through the blood brain barrier to interact with target dopamine receptors. Symptomatic treatment of Parkinson’s remains dopamine replacement, including the DA’s. Dopamine agonists are frequently the first line of choice for therapy for the just diagnosed Parkinson’s patient. Dopamine agonists do help control motor symptoms in Parkinson’s although there can be significant side-effects (see Table below). Also below is a Table describing DA’s. The DA side effects can become intolerable for some people-with-Parkinson’s, and the decision to taper or withdraw the DA is made. Or maybe you’re a candidate for deep-brain stimulation (DBS) surgery and to calibrate the device you’ll be asked to stop your Parkinson’s medication for a short period of time.
“I enjoy convalescence. It is the part that makes the illness worth while.” George Bernard Shaw
First report of dopamine agonist withdrawal syndrome (DAWS): Dopamine agonist withdrawal syndrome (DAWS) was first described in 2010 by Rabinak and Nirenberg on five of their patients with non-motor impulse control behavioral disorders (ICD) caused by the DA; thus, they were tapered. Two patients were further described in this publication. The first patient was a 67-year-old woman with a six year history of Parkinson’s, and she had been taking various drugs including a DA. She had developed a difficult ICD, and they elected to taper the DA; unexpectedly, she then had severe anxiety and dysphoria. They tried an increase in carbidopa/levodopa and they used other therapy for cognitive behavior control; to no benefit to the patient. They changed her back to the original DA dose and she had a rapid and dramatic improvement in all of her symptoms. This patient continues to use the DA and remains with the difficult ICD.
Patient #2 was a 61-year-old woman with a six-year history of Parkinson’s and likewise an ICD prompted by the DA; she began a DA tapering with increased carbidopa/levodopa medication. During the DA taper, she developed depression and severe anxiety and became agitated; she also had fatigue and insomnia. As with Patient #1, adding back the DA improved all of her non-motor symptoms. It took several years for her to successfully reduce her DA doseage. The figure below visually highlights some of the key symptoms of DAWS.

What both cases shared were prominent psychiatric symptoms, poor response to both additional carbidopa/levodopa (to take the place of the DA) and psychiatric medication; however, both had rapid improvement in their ‘new symptoms’ when placed back on the DA. The majority of DAWS symptoms are presented in the the Table below. “The secret of learning to be sick is this: Illness doesn’t make you less of what you were. You are still you.” Tony Snow
“The secret of learning to be sick is this: Illness doesn’t make you less of what you were. You are still you.” Tony Snow
Risk-factors and prevalence of DAWS: Since the original study in 2010, there have been several follow-up studies on DAWS. Some of the studies speculated that a large DA dose in the presence of pre-existing ICD are the most important risk factors for DAWS. The ‘number’ talked about frequently is something called the ‘levodopa equivalent daily dose’ (LEDD) of the dopamine agonist, where it has been suggested that >150 mg was linked to an increased risk of DAWS. Use this on-line program to calculate your LEDD (click here). Here is an LEDD example: someone taking 14 mg ropinirole/day (with the online algorithm), the LEDD would be 280 mg daily. What? OK, so what did you say? This means if you wanted to replace the 14 mg/day ropinirole with carbidopa/levodopa you would need about 300 mg per day of levodopa based on this calculation. I refer you to do the papers cited at the end of the blog post for more details about LEDD. What is interesting is several of the studies have compared the taper versus total withdrawal of the DA; it does not seem to alter the risk of DAWS. Good news is if you’re not having any detrimental side effects from the DA, just continue on and you’re good to go. The bad news is if you are having some side effects and you want to try and eliminate them by tapering down need to carefully consult with your neurologist and work up a feasible plan. Please remember I’m a biochemist, not a physician, and I just am interpreting data from publications.
The prevalence of DAWS has been reported to be between 15 and 19% in patients with Parkinson’s; it seems to be consistently about one-in-five. As mentioned previously, there appears to be no difference in relative risk of DAWS comparing patients that discontinue DA completely or those that reduce the DA by taper. Based on the percentage mentioned above, this says ~4 out of 5 people-with-Parkinson’s can DA taper without any problems.
“It is in moments of illness that we are compelled to recognize that we live not alone but chained to a creature of a different kingdom, whole worlds apart, who has no knowledge of us and by whom it is impossible to make ourselves understood: our body.” Marcel Proust
DA mechanism of action to cause DAWS: To recap, DAWS occurs in a subset of patients with Parkinson’s that have had difficulties managing the side effects of a DA, and the decision has been made to remove that DA from the patient’s regimen. The simplest notion is that you would then replace the DA with an increased dose of carbidopa/levodopa (using the LEDD); however, this is Parkinson’s and this is the brain and it’s just not going to be that easy. The diagram below summarizes a very simplistic view of dopamine and DA’s in their interactions with motor and reward pathways. There is no doubt that in treating Parkinson’s, the replacement of dopamine is crucial for many different physiological functions in the human body. Dopamine agonists and dopamine share similar binding properties to dopamine receptors. They are very important in improving motor symptoms (through the nigrostriatal pathway) but there is also some potential detrimental crossover to the reward center (through the mesocorticolimbic pathway). It is this minor pathway that is linked to the increased risk of ICD in some patients being treated with a DA. It is not clear, however from the data published so far that there is a difference in this 20% of the patient population in their mesocorticolimbic circuitry system with the DA in comparison to the other 80% of the population. In summary, what causes DAWS during DA tapering is not well understood.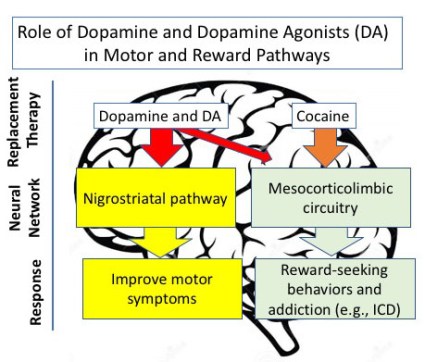
“Medicine is intention. Those who are proficient at using intention are good doctors.” Sun Simiao
Treatment/management of DAWS during DA taper: DAWS is a relatively recent phenomena related to DA withdrawal. Patients with (i) a predisposition to ICD and (ii) a larger dose of DA are apparently at increased risk of developing DAWS. There is no well-delineated treatment plan that the neurologist can follow; best recommendation (from the papers cited below) is the patient should be tapered at a very slow dose reduction over a long period of time, and see what happens. Clearly, it is crucial that the patient and the neurologist carefully evaluate signs of ICD and DAWS at every visit, especially for patients at high risk.
“The treatments themselves do not ‘cure’ the condition, they simply restore the body’s self-healing ability.” Leon Chaitow
Summary: As someone with Parkinson’s, I’ve done a lot of reading about treatment strategies (what’s good and what’s not so good). For someone my age there would almost always be a recommendation to begin the DA (the so-called sparing one of levodopa until it’s absolutely needed) and then as symptoms progressed, you would switch over and combine the DA with carbodipa/levodopa. Had I read the opinions of Dr. Ahlskog in the beginning, I might have opted to start with carbidopa/levodopa without the DA (Ahlskog JE. Cheaper, Simpler, and Better: Tips for Treating Seniors With Parkinson Disease. Mayo Clinic Proceedings. 2011;86(12):1211-6. doi: https://doi.org/10.4065/mcp.2011.0443). Biochemically, DAWS is an interesting problem but there needs to be additional studies to delineate the mechanism of action. Finally DAWS clinically is worrisome and definitely not well-understood; and likely, the scope of DAWS is under-recognized.
Key References:
- Rabinak CA, Nirenberg MJ. Dopamine agonist withdrawal syndrome in Parkinson disease. Arch Neurol. 2010;67(1):58-63. doi: 10.1001/archneurol.2009.294. PubMed PMID: 20065130.
- Nirenberg MJ. Dopamine agonist withdrawal syndrome and non-motor symptoms after Parkinson’s disease surgery. Brain. 2010;133(11):e155; author reply e6. doi: 10.1093/brain/awq165. PubMed PMID: 20659959.
- Cunnington AL, White L, Hood K. Identification of possible risk factors for the development of dopamine agonist withdrawal syndrome in Parkinson’s disease. Parkinsonism Relat Disord. 2012;18(9):1051-2. doi: 10.1016/j.parkreldis.2012.05.012. PubMed PMID: 22677468.
- Pondal M, Marras C, Miyasaki J, Moro E, Armstrong MJ, Strafella AP, Shah BB, Fox S, Prashanth LK, Phielipp N, Lang AE. Clinical features of dopamine agonist withdrawal syndrome in a movement disorders clinic. J Neurol Neurosurg Psychiatry. 2013;84(2):130-5. doi: 10.1136/jnnp-2012-302684. PubMed PMID: 22933817.
- Edwards MJ. Dopamine agonist withdrawal syndrome (DAWS): perils of flicking the dopamine ‘switch’. J Neurol Neurosurg Psychiatry. 2013;84(2):120. doi: 10.1136/jnnp-2012-303570. PubMed PMID: 22993451.
- Nirenberg MJ. Dopamine agonist withdrawal syndrome: implications for patient care. Drugs Aging. 2013;30(8):587-92. doi: 10.1007/s40266-013-0090-z. PubMed PMID: 23686524.1.
- Nirenberg MJ. Dopamine agonist withdrawal syndrome: implications for patient care. Drugs Aging. 2013;30(8):587-92. doi: 10.1007/s40266-013-0090-z. PubMed PMID: 23686524.
- Solla P, Fasano A, Cannas A, Mulas CS, Marrosu MG, Lang AE, Marrosu F. Dopamine agonist withdrawal syndrome (DAWS) symptoms in Parkinson’s disease patients treated with levodopa-carbidopa intestinal gel infusion. Parkinsonism Relat Disord. 2015;21(8):968-71. doi: 10.1016/j.parkreldis.2015.05.018. PubMed PMID: 26071817.
- Huynh NT, Sid-Otmane L, Panisset M, Huot P. A Man With Persistent Dopamine Agonist Withdrawal Syndrome After 7 Years Being Off Dopamine Agonists. Can J Neurol Sci. 2016;43(6):859-60. doi: 10.1017/cjn.2015.389. PubMed PMID: 26842385.
- Patel S, Garcia X, Mohammad ME, Yu XX, Vlastaris K, O’Donnell K, Sutton K, Fernandez HH. Dopamine agonist withdrawal syndrome (DAWS) in a tertiary Parkinson disease treatment center. J Neurol Sci. 2017;379:308-11. doi: 10.1016/j.jns.2017.06.022. PubMed PMID: 28716269.
- Yu XX, Fernandez HH. Dopamine agonist withdrawal syndrome: A comprehensive review. J Neurol Sci. 2017;374:53-5. doi: 10.1016/j.jns.2016.12.070. PubMed PMID: 28104232.
- Solla P, Fasano A, Cannas A, Marrosu F. Dopamine agonist withdrawal syndrome in Parkinson’s disease. J Neurol Sci. 2017;382:47-8. doi: 10.1016/j.jns.2017.08.3263. PubMed PMID: 29111017.
“Life always gives us exactly the teacher we need at every moment. This includes every mosquito, every misfortune, every red light, every traffic jam, every obnoxious supervisor (or employee), every illness, every loss, every moment of joy or depression, every addiction, every piece of garbage, every breath. Every moment is the guru.” Joko Beck
Cover photo credit: f.fwallpapers.com/images/sun-peeking-through-snow-covered-trees.jpg
Agitation- img.aws.livestrongcdn.com/ls-article-image-400/cme/cme_public_images/www_livestrong_com/photos.demandstudios.com/49/85/fotolia_4199215_XS.jpg
Depression- http://www.scientificamerican.com/sciam/cache/file/FCD288AE-5C2E-49F2-85858FA255A8034B_source.jpg
Fatigued- www.belmarrahealth.com/wp-content/uploads/2017/03/fatigue-in-the-elderly-300×200.jpg
Panic attack- lifetimewoman.com/wp-content/uploads/2016/09/panica-1.jpg


What is your take on when alpha-synucleun, nilotinib, or something of a “cure” will be available in the PD market?
LikeLike