“Any fool can know. The point is to understand.” Albert Einstein
“To acquire knowledge, one must study; but to acquire wisdom, one must observe.” Marilyn vos Savant
Introduction: I have used withering grapes on a grape vine to depict dopaminergic cells in Parkinson’s disease. It is generally thought that a substantial amount of dopaminergic cells (>50-60%) must be lost for the motor symptoms to become apparent. Would altering the amount of dopamine produced or increasing dopamine receptors in someone with Parkinson’s allow an individual to approach managing their disease differently or more effectively?
“Beware of false knowledge; it is more dangerous than ignorance.” George Bernard Shaw
Citicoline (CDP-Choline), Omega-3 Fatty Acids, and Caffeine: Parkinson’s is an evil disease; no doubt it works endlessly to make our lives less than suitable. It seems to start, much of the time, as a nuisance, yet over the years, it builds up and begins to dominate our brains, our bodies, and our quality of life. We replace the missing dopamine with levodopa/carbidopa derivatives, dopamine agonists, substances that prolong levodopa’s half-life, and substances that prolong dopamine’s action, and we undergo surgery to stimulate parts of our brain, which, magically, reduces the severity of some of our symptoms.
None of the treatments described above alters the disease trajectory. However, we are learning that strenuous exercise can be neuroprotective and, over time, induce neuroplasticity. We have an arsenal of supplements, natural chemical compounds, and essential vitamins/minerals that replace elements consumed by the disease.
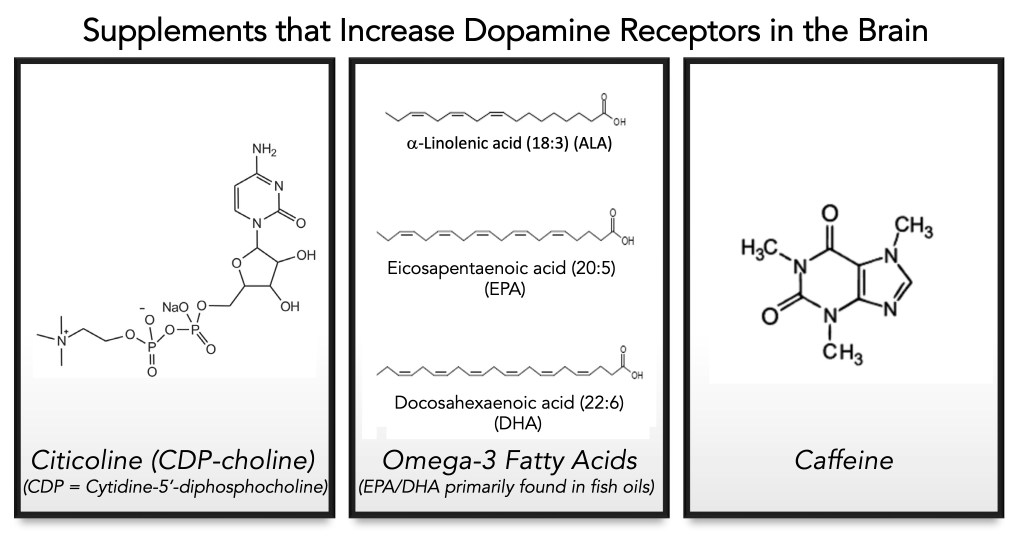
The focus of this blog post is the potential use of supplements that augment the dopaminergic machinery itself, which has the promise of modifying treatment and potentially slowing progression by maintaining or even increasing dopamine levels or altering the action or number of dopamine receptors. Ultimately, the potential of these supplements could boost the dopamine system and require less levodopa in an individual with Parkinson’s than without the use of these supplements. These compounds are shown below and will be discussed in the order of citicoline (CDP-choline), omega-3 fatty acids, and caffeine.
“The beautiful thing about learning is nobody can take it away from you.” B. B. King
Citicoline (CDP-choline)
In 1982, Agnoli et al. conducted a study in patients with Parkinson’s disease on carbidopa/levodopa, treating one group with 500 mg CDP-choline and the other with a placebo. They tested changes in tremor, rigidity, and bradykinesia. The group treated with CDP-choline showed improvement in rigidity and bradykinesia, and they speculate that changes in dopamine receptors on neuronal cells were supported by CDP-choline treatment.
Moving to 2020, Que et al. reviewed 7 clinical studies of CDP-choline in Parkinson’s. In this review, they found significant improvements in rigidity, akinesia, tremor, handwriting, and speech in the CDP-choline group. Interestingly, the use of CDP-choline reduced the effective levodopa dose required in patients.
In 2021, Nakazaki et al., found that treatment with 500 mg/day of CDP-choline, improved the memory function in older adults. They also reported that treatment with 1 gram/day of CD-choline had no adverse effects during the 12-week study.
What is the function of CDP -choline in the brain? CDP-choline is known to increase the synthesis of phosphatidylcholine, the primary phospholipid in neuronal membranes. Phosphatidylcholine is required for cell membrane integrity, proper function, and repair. And with age, phosphatidylcholine vesicles are reduced. Furthermore, CDP-choline can enhance the interaction between acetylcholine, norepinephrine, and dopamine. Specifically, it has been reported that CDP-choline improves dopamine synthesis, protects dopaminergic neurons, and can apparently restore functional dopamine receptors in aging. Importantly, CDP-choline has been shown to activate the cholinergic system, thereby indirectly altering dopamine and dopaminergic receptor activity.
“The love of knowledge is a kind of madness.” C. S. Lewis
Are there any downsides to taking citocoline (CDP-choline)? Many references state that the supplement is safe to use. By contrast, there were a series of articles posted on HealthUnlocked, authored by a very well respected member of this community, arguing the possibility of CDP-choline being broken down to a more toxic derivative (trimethylamine) and the unusual impact on this individual’s levodopa/carbidopa levels that increased after stopping the use of CDP-choline, and include these topics: “Citicoline: patients treated with citicoline experienced a significant worsening 45 days after the medication was discontinued. Updated” (click here), “Citicoline Warning” (click here), and “Citicoline in Parkinson’s Disease” (click here). However, it makes sense to measure the level of trimethylamine oxide in your blood (Trimethylamine N-oxide is a gut-microbiota-dependent metabolite linked to increased risks of heart attack, stroke, and cardiovascular death) to ensure that using CDP-choline does not raise levels of this cardiotoxin.
“The love of knowledge is a kind of madness.” C. S. Lewis
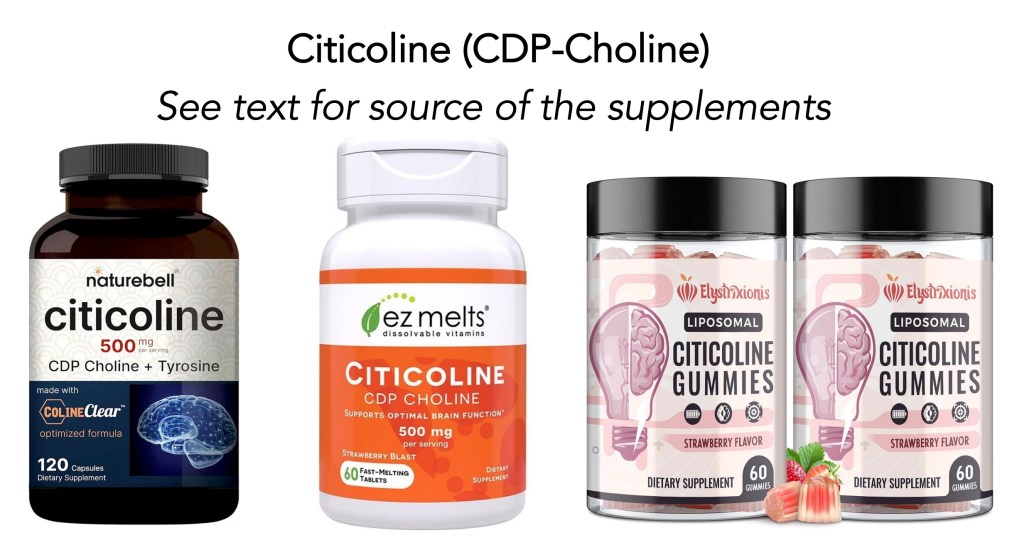
My History with CDP-Choline: I have tried it twice over the past 2 years. In 2024, I started with 500 mg/day of CDP-choline (2 capsules in the morning using NatureBell Citicoline (Amazon.com, click here). After 2 months of using it, I was taking about 200 mg less of levodopa/carbidopa (i..e, generic form of Sinemet) than the 1300 mg/day that I had been taking. One knee operation, and then switching to Crexont, and preparing for the first hip replacement surgery, led me to simplify my supplement schedule, and I stopped using it.
Following the switch to Crexont, a whole new array of issues emerged. Although Crexont definitely works as advertised, it comes with a ‘penalty’ (well, that’s what I call it). The relative dose between Crexont and “old-fashioned” carbidopa/levodopa, for me, my Neurologist had me taking 700 mg/dose of levodopa in Crexont compared to 250 mg/dose of standard levodopa. Whatever the cause (my age, my length of time taking levodopa), I developed levodopa-induced dyskinesia using Crexont. So, for me, Crexont increased the levodopa ‘on’ time before the ‘off’ time; however, the beneficial effect was counterbalanced by the adverse effect of levodopa-induced dyskinesia.
What to do? I started reading the literature, and as usual, developed the following plan. Phase 1: Go back and try CDPcholine again. Phase 2: Add omega-3 fish oil (fatty acids). I started using EZ Melts Citicoline [500 mg/day dissolving tablets (click here). After a month or so, I switched to another form, taking the Citicoline Supplement from Elystrixionis (2 gummies per day, providing 1000 mg of CDP-choline) and fish oil (see the story below this one for the details). After a few months, I started tapering my Crexont. See this blog post for the details, “Journey with Parkinson’s: ‘Fine-tuning’ Dopaminergic Therapy” (click here). It took about 6 weeks, but I was able to reduce my Crexont levodopa dose from 2800 mg per day to 2520 mg per day.
This may not seem like a big difference, but it dramatically reduced my dyskinesia, and the levodopa drop-off is essentially the same. Of course, the question now is: was this change due to the dopaminergic machinery in my brain changing with CDP-choline and fish oil? Or just me refining my body’s requirement of the mega-doses of levodopa found in Crexont? The only way I can answer that is to taper off the supplements and see if my brain requires more Levodopa (Crexont) to maintain my current brain/body state. Regardless, it helps me substantially, and if it is real, with time, I can work up to a higher dose of Crexont as needed. Until then, I am just keeping both supplements going (and as usual, my n = 1 experiment is terribly flawed).
“Knowledge is an excellent drug; but no drug has virtue enough to preserve itself from corruption and decay, if the vessel be tainted and impure wherein it is put to keep.” Michel de Montaigne
Omega-3 Fatty Acids (Fish Oils)
We always think that a big cause of our weight gain as we age is fat (also known as lipids). Please remember that every cell in our body, every cell, requires lipids for the functioning and fluidity of the cell membrane. Thus, lipids/fats, whatever we call them, are absolutely essential to our existence.
Yes, we do have good lipids and bad lipids. And yes, consuming bad lipids will contribute to morbidity in the human condition. The focus here is centered around some good lipids, the omega-3fatty acids (lipids), pictured at the beginning of the blog. Lipids are different in structure and nature from proteins and carbohydrates. They usually contain carbon, hydrogen, and oxygen and are not water-soluble (as are many proteins and carbohydrates). We place fatty acids into two main types: saturated and unsaturated. Unsaturated fats are further broken down into monounsaturated and polyunsaturated fats. Omega-3 fatty acids are polyunsaturated fatty acids; they are essential for life. We do not make these lipids; they must be obtained from food sources or as supplements.
There are three main types of omega-3 fatty acids:
alpha-linolenic acid (ALA)
eicosapentaenoic acid (EPA)
docosahexaenoic acid (DHA)
We get most of our omega-3 fatty acids from our diets; thus, eating fish and plants is a rich natural source. Omega-3 fatty acids are abundant in salmon, anchovies, herrings, mackerel, and tuna, to name a few, and in flaxseed, canola oil, edamame, soybeans, and walnuts as plant sources. Of course, there are many reasons someone may not eat fish, such as being allergic or following a vegetarian (or vegan) diet. Thus, the development of fish oil supplements. Numerous clinical trials have shown that omega-3 fatty acids can lower your risk of cardiovascular disease, and beyond heart disease, omega-3 fatty acids may also reduce your risk of some cancers, neurodegenerative diseases, and age-related macular degeneration.
“Thinking is more interesting than knowing, but less interesting than looking.” Johann Wolfgang von Goethe
The Impact of Omega-3 Fatty Acids on Dopaminergic Neuroransmitter, Dopamine Receptors, and Parkinson’s: As with many disorders, deficiencies in nutrients can have a detrimental impact on the test animal. Delion et al., in 1996, studied the impact of a diet deficient in alpha-linolenic acid (ALA) in an animal model. Rats fed diets deficient in ALA showed severe deficiencies in dopaminergic and serotonergic neurotransmitters in the striatum, frontal cortex, and cerebellum. They also found a reduction in dopaminergic D2 receptors.
By contrast, in 1998, Chalon et al fed rats a diet enriched with fish oils (salmon and palm oil, each at 50%) and compared to diets enriched wth peanut oil and rapeseed oil. Rats fed the salmon/palm oil mixture showed an increase of 40% dopamine in the frontal cortex, which also showed a reduction in monoamine oxidase B and greater dopamine binding to dopamine D2 receptors. Interestingly, there was a reduction in dopamine binding to dopamine D2 receptors in the striatum.
Recently, Detopoulou et al. reviewed omega-3 fatty acid supplementation in the diets of patients with Parkinson’s disease. PwP have lower levels of 3-omega fatty acids compared to age-matched control patients without Parkinson’s. Through these studies, omega-3 fatty acids counteract neuroinflammation, promote neuronal survival, and can promote neuroplasticity. In conclusion, the use of 1000 mg of omega-3 fatty acids in a human diet shows much promise in delaying the progression of Parkinson’s. As additional clinical studies examining omega-3 fatty acids are conducted, there is a strong likelihood that supplementation will remain a useful approach for addressing the consequences of Parkinson’s.
“Few can tell what they know without also showing what they do not know.” Ivan Panin
My History with Omega-3 Fatty Acids: After reading about the benefits of omega-3 fatty acids in Parkinson’s, it seemed logical to add them along with the CDP-choline supplement and see what happens. I have tried two different fish oil formulas. The first was Qunol Fish Oil Omega-3 Mini Softgels (click here), which provided 1000 mg of omega-3 EPA (645 mg) and DHA (310 mg), and 45 mg of other omega-3 fatty acids. I have always had a pill/capsule size and shape limit. Usually, anything >600 mg is hard for me to swallow. And the type of supplement matters as well; capsules are usually better than tablets. I thought these mini softgels would be okay, but they were constantly getting stuck on the way down. I have switched to LiliCare Fish Oil Omega-3 Gummies 1500 mg for Adults – Triple Strength with EPA (800 mg) and DHA (400 mg) (click here). The results described above are what I feel the CDP-choline and omega-3 fatty acids are contributing to the improved microenvironment in my dopamine-producing area. As noted before, I have only the change noted in the amount of Crexont I take on a daily basis. Regardless, the studies performed to date all suggest a beneficial role for using omega-3 fatty acids as supplements.

“Education is an admirable thing, but it is well to remember from time to time that nothing that is worth knowing can be taught.” Oscar Wilde
Caffeine:
The most consumed psychoactive substance is caffeine. There are many caffeine-containing beverages and foods. Caffeine has long been considered to be neuroprotective for Parkinson’s, but it may also have benefits to both motor and non-motor components of the disorder. Several large studies have clearly demonstrated that caffeine consumption decreases the risk of developing Parkinson’s. For me, I have been a life-long consumer of a relatively large amount of caffeine each day since my college years (drinking black coffee was the ‘curse’ of all-night study sessions for Organic Chemistry!).
The effect of caffeine in Parkinson’s disease is largely mediated by antagonizing brain adenosine A2A receptors, thereby conferring neuroprotection and modulating mitochondrial excitotoxicity. Interestingly, Volkow et al. showed that a 300 mg dose of caffeine (comparable to 2-3 cups of coffee) increased the availability of dopamine D2/D3 receptors in the putamen and ventral striatum. Whether this was an increase in the dopamine effect or a potential increase in affinity, this result suggests that caffeine, when consumed in relatively large amounts, may require less dopamine function, or, as done here, to measure alertness. How would the use of coffee translate to treating Parkinson’s? It would be difficult, but there is an A2A receptor antagonist drug, istradefylline, for the treatment of “off” time effects of levodopa.
Cummins and Gates reviewed the use of Istradefylline in Parkinson’s (Cummins L, Cates ME. Istradefylline: A novel agent in the treatment of “off” episodes associated with levodopa/carbidopa use in Parkinson disease. Mental Health Clinician. 2021 Dec;12(1):32–36. doi:10.9740/mhc.2022.01.032); they concluded: “Istradefylline was recently approved by the FDA for treatment of PD. In randomized, double-blind, placebo-controlled studies, istradefylline decreased “off” time in patients taking L/C, and it was well tolerated. Its unique pharmacology includes antagonism of the adenosine A2A receptor and lack of direct dopaminergic effects, which is potentially advantageous relative to other drugs that can be selected to treat patients with PD. However, the cost of therapy could be prohibitive in certain circumstances. Real-world use and additional research will further define its role in treatment of PD.”
“Ultimately, there is no such thing as failure. There are lessons learned in different ways.” Twyla Tharp
Here are two scenarios to consider: would CDP-choline and omega-3 fatty acids help?:
#1.) What if you are relatively young and have just been informed of your Parkinson’s: could you slow down the progression, try to repair the damaged neurons, and preserve the dopamine- generating neurons while you are still relatively young? Could you alter the neurons in this toxic brain microenvironment to make more dopamine receptors available? What then would be the action regarding this individual with Parkinson’s? Would they require less dopaminergic therapy in absolute amount, and would this allow them to be treated longer and more effectively?
#2.) The individual with Parkinson’s has had the disease for at least a decade and has fewer functional dopaminergic cells than those found in scenario #1. This patient requires substantially more carbidopa-levodopa in terms of amount and frequency of use. Chronic treatment with carbidopa/levodopa has finally led to the development of motor complications, wearing-off issues, and dyskinesia in most patients with Parkinson’s. The extremes of the ‘On-Off’ can produce a levodopa-induced dyskinesia during the ‘on’ phase and then a crippling Parkinsonism [including the really cruel freezing of gait (FOG)] during the ‘off’ phase. Yes, I was starting to feel the consequences of the on-off phase of my carbidopa/levodopa, as FOG would occur if I allowed the Sinemet to bottom out (see my post about FOG).
Certainly, I believe that the younger, almost entry-level Parkinson’s patient (described as patient #1) would benefit from using CDP-choline and omega-3 fatty acids. Anything that could lead to a healthier and more proficient substantia nigra (pars compacta and pars reticulata), and the other components for motor activity, striatum (caudate and putamen), globus pallidus (internal and external segments), and thalamus would allow this PwP a much better scenario to maintain a high quality of life.
In patent #2, an older, more-established Parkinson’s patient, with a substantial burden of carbidopa/levodopa, and elements of on-off difficulties, certainly shifting the burden to require less levodopa certainly sounds hopeful for this person. I certainly believe it has helped me by taking CDP-choline and omega-3 fatty acids while taking Crexont.
However, for some individuals with Parkinson’s, they might not react this way to these supplements. The progressive and sustained exposure of the nigrostriatal machinery places this region of the brain in a somewhat different physiological state. Thus, the burden of long-term carbidopa/levodopa treatment is detrimental to the dopamine-focused machinery, and it might not respond as well to these supplements as did the younger patient described above. I refer you to the review by Riederer et al. (2025), which reviewed the use of levodopa in Parkinson’s from the early to the later, more advanced stages of the disease.
“You won’t learn if you think you already know everything.” Robert Kiyosaki
A Final Thought: Wherever you are on the spectrum of progression of Parkinson’s, how you manage your disorder depends on your awareness and perception. These are your observations and feelings, not your Neurologist (because they are only getting a linear response to questions every six months). When you ask your CarePartner, family, and friends about the progression of ‘your’ Parkinson’s, when you think deeply about managing your quality of life, then and only then, can you decide which path you want to follow. That path will be the right one for you, and that is what matters the most. There is no right answer to using supplements; there’s only the right decision for you to make after educating yourself. Good luck in your journey, and I look forward to our paths crossing again in the near future.
Medical Disclaimer:
This blog post contains information related to health care. It is not intended to replace medical advice. It should be used to supplement, rather than replace, your neurologist’s regular care. It is recommended that you seek your physician’s advice/approval before embarking on any new health plan or changes to your existing plan/routine.
Please note that the views and opinions expressed here are my own. I am not a physician, and this content is presented for informational purposes only and not as medical advice. Just because I take these supplements does not prove their safety and efficacy. Please consult your neurologist/physician if you are interested in this material.

References
Citicoline (CDP-choline):
Arenth, Patricia M., Kathryn C. Russell, Joseph H. Ricker, and Ross D. Zafonte. “CDP-choline as a biological supplement during neurorecovery: a focused review.” PM&R 3, no. 6 (2011): S123-S131.
Nakazaki, Eri, Eunice Mah, Kristen Sanoshy, Danielle Citrolo, and Fumiko Watanabe. “Citicoline and memory function in healthy older adults: a randomized, double-blind, placebo-controlled clinical trial.” The Journal of nutrition 151, no. 8 (2021): 2153-2160.
Que, Diana-Lynn S., and Roland Dominic G. Jamora. “Citicoline as adjuvant therapy in parkinson’s disease: A systematic review.” Clinical therapeutics 43, no. 1 (2021): e19-e31.
Agnoli, A., S. Ruggieri, A. Denaro, and Giuseppe Bruno. “New strategies in the management of Parkinson’s disease: a biological approach using a phospholipid precursor (CDP-choline).” Neuropsychobiology 8, no. 6 (1982): 289-296.
Omega-3 Fatty Acids (Fish Oils):
Avallone, Rossella, Giovanni Vitale, and Marco Bertolotti. “Omega-3 fatty acids and neurodegenerative diseases: new evidence in clinical trials.” International journal of molecular sciences 20, no. 17 (2019): 4256.
Detopoulou, Paraskevi, Gavriela Voulgaridou, Alexandra Saridaki, Elissaios-Minos Argyris, Vasiliki Seva, Vasilios Dedes, Constantinos Giaginis, Georgios I. Panoutsopoulos, and Sousana K. Papadopoulou. “Omega-3 fatty acids’ supplementation in Parkinson’s disease: A systematic review of randomized controlled trials.” Clinical Nutrition Open Science 55 (2024): 102-115.
Delion, Sylvie, Sylvie Chalon, Denis Guilloteau, Jean‐Claude Besnard, and Georges Durand. “α‐Linolenic acid dietary deficiency alters age‐related changes of dopaminergic and serotoninergic neurotransmission in the rat frontal cortex.” Journal of neurochemistry 66, no. 4 (1996): 1582-1591.
Chalon, Sylvie, Sylvie Delion-Vancassel, Catherine Belzung, Denis Guilloteau, Anne-Marie Leguisquet, Jean-Claude Besnard, and Georges Durand. “Dietary fish oil affects monoaminergic neurotransmission and behavior in rats.” The Journal of nutrition 128, no. 12 (1998): 2512-2519.
Caffeine:
Ren, Xiangpeng, and Jiang-Fan Chen. “Caffeine and Parkinson’s disease: multiple benefits and emerging mechanisms.” Frontiers in Neuroscience 14 (2020): 602697.
Prediger, Rui DS. “Effects of caffeine in Parkinson’s disease: from neuroprotection to the management of motor and non-motor symptoms.” Journal of Alzheimer’s disease 20, no. s1 (2010): S205-S220.
Volkow, Nora D., Gene-Jack Wang, Jean Logan, David Alexoff, Joanna S. Fowler, Panayotis K. Thanos, C. Wong, Vicent Casado, Sergi Ferre, and Dardo Tomasi. “Caffeine increases striatal dopamine D2/D3 receptor availability in the human brain.” Translational psychiatry 5, no. 4 (2015): e549-e549.
Cummins, Lauren, and Marshall E. Cates. “Istradefylline: A novel agent in the treatment of “off” episodes associated with levodopa/carbidopa use in Parkinson disease.” Mental Health Clinician 12, no. 1 (2022): 32-36.
Levodopa:
Riederer, Peter, Sabrina Strobel, Toshiharu Nagatsu, Hirohisa Watanabe, Xiqun Chen, Peter-Andreas Löschmann, Jeswinder Sian-Hulsmann et al. “Levodopa treatment: impacts and mechanisms throughout Parkinson’s disease progression: P. Riederer et al.” Journal of Neural Transmission 132, no. 6 (2025): 743-779.
Cover Photo Image by Łukasz Tekieli from Pixabays