“Our destiny is not determined by the number of times we stumble but by the number of times we rise up, dust ourselves off, and move forward.” Dieter F. Uchtdorf
“My motto was always to keep swinging. Whether I was in a slump or feeling bad or having trouble off the field, the only thing to do was keep swinging.” Hank Aaron
Introduction: A few months ago, a friend and reader of this blog asked me if I had ever heard of palmitoylethanolamine (usually abbreviated as PEA). I said no but that I would read about it, report on it, and if I saw some potential in its use for Parkinson’s, I would obtain some of it and see how it works. [Please NOTE: Several references are listed at the bottom of this blog post.]
I was further referred to an active dialog about the use of PEA by several notable persons who use Cure Parkinson’s at the Health Unlocked site (https://healthunlocked.com/cure-parkinsons) as their forum. Over the years, I have found this to be a helpful website regarding many different topics on Parkinson’s, with comments by some truly remarkable people.
The current blog post will describe some of the uses of PEA in neurological disorders, how PEA controls neuroinflammation, the different forms of PEA, and the successful use of PEA to treat the motor and non-motor symptoms of Parkinson’s clinically. But I want to begin with an overview of inflammation and then segue into the biochemistry and biological properties of PEA..
“It’s easier to go down a hill than up it but the view is much better at the top.” Henry Ward Beecher
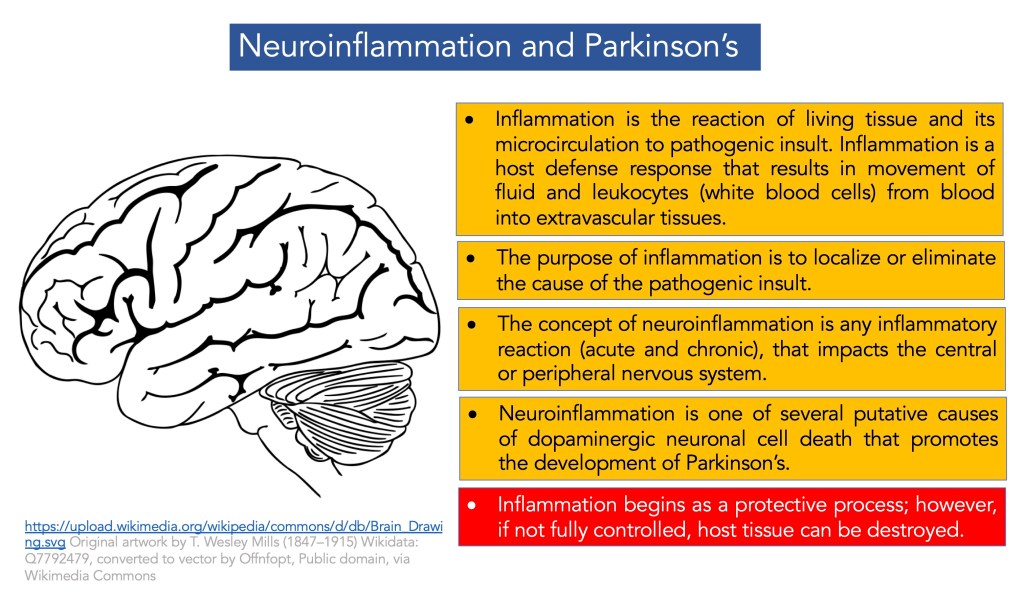
Introduction to Inflammation: In is the body’s host defense response to some pathogenic insult or substance. There are three main aspects to mention regarding the definition of inflammation:
- Inflammation is the reaction of living tissue and its microcirculation to pathogenic insult.
- Generation of inflammatory mediators.
- Movement of fluid and leukocytes from blood into extravascular tissues.
The purpose of inflammation resides in four actions, namely:
- Localize or eliminate the cause of the pathogenic insult.
- Limit tissue injury.
- Remove or repair injured tissue components.
- Restore normal physiology.
Inflammation is usually beneficial, although it can cause substantial morbidity and mortality if left uncontrolled. The schematic below summarizes inflammation.
“Only those who will risk going too far can possibly find out how far one can go.” T. S. Eliot
Essential Concepts in Inflammation (NOTE: This section gives much more depth to understanding the body’s response to pathogenic substances. If you want to learn more about inflammation, please read on. If not, skip ahead because the rest of the post describes the role of PEA in regulating neuroinflammation and, specifically, Parkinson’s.). LIsted below are several fundamental concepts regarding the host defense response called inflammation.
- Inflammation is subdivided into acute and chronic based on the time course and the cellular components involved in the inflammatory response.
- Acute inflammation is caused by: Microbial infections, Tissue necrosis; Physical agents; Chemical irritants; Immune-mediated hypersensitivity.
- Chronic inflammation is caused by: Persistent tissue injury and acute inflammation; Microorganisms resistant to phagocytosis or intracellular killing (e.g., Mycobacteria, viruses, fungi, some parasites); Foreign bodies; Autoimmune disorders; Primary granulomatous diseases.
- Significant Manifestations of Acute Inflammation: Rapid vascular response with altered microvasculature; Increased vascular permeability with an accumulation of fluid and plasma components; Intravascular stimulation of platelets; Interaction of chemical mediators; Margination, adhesion, migration, and chemotaxis of neutrophils; Phagocytosis and intracellular killing.
- Acute inflammation is characterized by increased blood flow and vessel permeability, which leads to the extravasation of excess fluid (inflammatory edema).
- Activated oxygen species production accompanies phagocytosis: Superoxide anion (O2–); Hydrogen peroxide (H2O2 ); Hypochlorous acid (HOCl); Hydroxyl radical (.OH).
- Acute inflammation is characterized by interstitial edema and an influx of predominantly neutrophils. Chronic inflammation is characterized by interstitial fibrosis and the influx of predominantly mononuclear leukocytes, e.g., lymphocytes, monocytes, macrophages, and plasma cells.
- Chronic inflammation is induced: Stimulus is not readily degradable, for example, suture granuloma; stimulus is within a cell, for example, viral myocarditis; stimulus is a cell, for example, a fungus; persistent or recurrent injury, for example, gastric ulcer; induced by a T lymphocyte-mediated immune response, for example, contact dermatitis.
- Granulomatous inflammation is a particular form of chronic inflammation characterized by granuloma formation. A granuloma is a dense accumulation of predominantly mononuclear phagocytes, typically surrounded by a collar of lymphocytes.
- Without the proper response and control steps, inflammation will get out of control and can frequently cause much morbidity/mortality.
“It always seems impossible until it’s done.” Nelson Mandela
Palmitoylethanolamide Structure, Occurrence in Nature, and Biological Action: Palmitoylethanolamide (or PEA) is a bioactive lipid synthesized in the cell lipid bilayer in most human tissues, including the brain. The chemical structure of PEA is given below in the schematic.
Interestingly, PEA is found to be synthesized by many tissues in response to cellular injury; notably, PEA is upregulated in disease states. The broad action of PEA in responding to various diseases includes anti-inflammatory, analgesic, immunomodulatory, and neuroprotective. By saying this, one must wonder; it does everything; it is a ‘magic pill.’ Therein lies the difficulty with such a substance with a broad range of physiological responses. Is there truly one action that trumps all the others?
The answer to the best physiological role for PEA lies in its actions on biological systems and cell types. When studied with a diverse array of receptor and cell-based assay systems, the following results are given below.
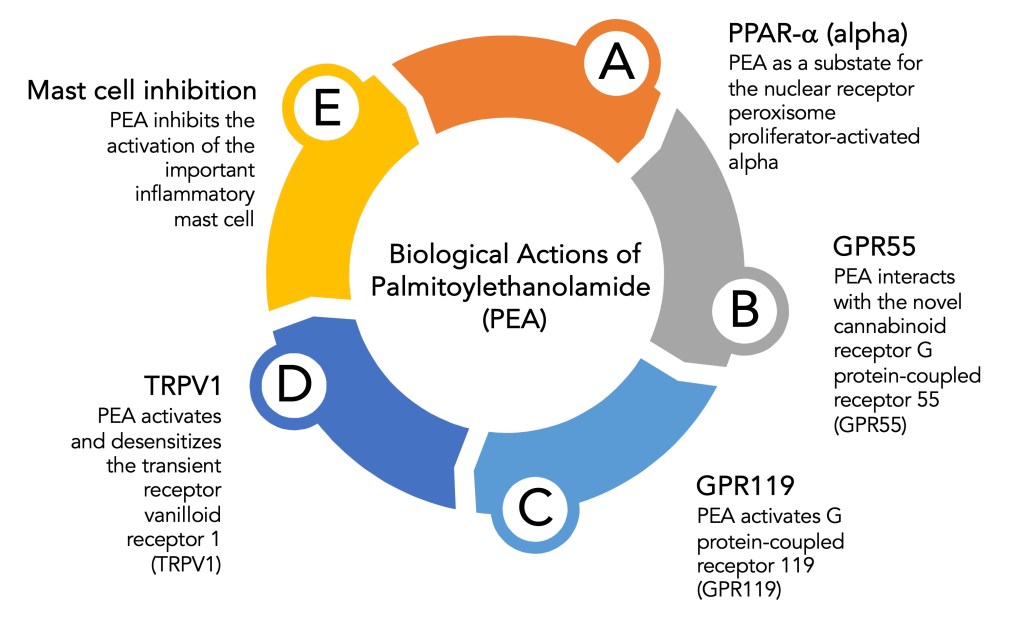
Due to the broad physiological targets found for PEA, this compound has been widely studied in many disorders, including the flu, allergies, chronic pain, psychological disorders, joint pain, antibacterial and antiviral activities, sleep disorders, and neurodegeneration. Furthermore, in animal and human studies, it is firmly established that PEA is an immunomodulating agent.
Neuroinflammation is one of the likely causes of Alzheimer’s, stroke, and traumatic brain injury (TBI). So yes, neuroinflammation is also one of the critical causes of Parkinson’s. Remember, earlier, I said that inflammation, including neuroinflammation, is a body’s response to a pathogenic substance. And in the beginning, the response starts with a protective goal. However, there are some circumstances where the reaction was too robust in the down-regulation of inflammation/neuroinflammation. This leads to injury and potentially tissue death or, in the least, altered function. Thus, a pleiotropic (responsible for or affecting more than one characteristic modification/change) substance like PEA has real potential for use in many neurodegenerative disorders.
“When obstacles arise, you change your direction to reach your goal; you do not change your decision to get there.” Zig Ziglar
Smaller and Very Small Forms of PEA: Here is a problem with PEA. It is a fatty acid amide, which means it is not a water-loving substance. We are a very hydrated and water-friendly entity. PEA is a lipid that does not dissolve in most things we eat, drink, or love to eat/drink. Thus, this has led scientists to get out their processing units and slice-and-dice PEA into more minor constituents. To increase the ‘bioavailability’ of the generally large particle and naturally a lipid by nature, they created micronized and ultra-micronized PEA. The good news is that while PEA is still not greatly water-soluble, the effect of micronizing PEA did not harm its biological properties; however, it was more effective at lower doses. Therefore, before you rush out and order something from Amazon.com, please note that ultra-micronized PEA and micronized PEA > non-modified PEA are more effective; that is to say, the smaller size, the PEA product you may be using one day appears to be better if micronized to some extent.
“Try and fail, but don’t fail to try.” John Quincy Adams
Ultra-micronized Palmitoylethanolamide (um-PEA) as Therapy for Parkinson’s: Until now, I have only hinted that PEA has the potential to improve the major motor and non-motor symptoms of Parkinson’s. In 2017, yes, I said 2017, a Movement Disorder group in Florence, Italy, examined 30 patients with Parkinson’s in what they described as a prospective observational study for 15 months. This work was performed by Dr. Stefania Brotini and associates.

The following points represent this fascinating study:
•The patients tested in this clinical investigation were 73 years of age on average. They were diagnosed using the Parkinson’s Disease Society Brain Bank clinical diagnostic criteria, and they were all taking levodopa at the study’s time. Patients with other forms of parkinsonism were excluded from the study.
•To evaluate motor and non-motor symptoms, they used the revised Movement Disorder Society/Unified Parkinson’s Disease Rating Scale (MDS-UPDRS).
•MDS-UPDRS clinical assessment was carried out monthly for three consecutive months before the actual trial began (times -3,-2,-1, 0). After that, um-PEA treatment (Normast, a medical food product available in Italy)) at a dose of 600 mg, two sublingual sachets for a total of 1200 mg um-PEA/day, were added to levodopa therapy (and eventually to other PD medication) for three months, followed by one sachet/day (total of 600 mg/day of um-PEA) for up to 12 months. Patients underwent MDS-UPDRS clinical assessment during um-PEA treatment at months 1, 3, 6, and 12.
•Non-motor experiences of daily living (nM-EDL): There was a significant improvement in the nM-EDL scores, going from 9.7 ± 1.18 at the start to 4.5 ± 0.69 at one year with um-PEA adjuvant therapy. Most notably, a significant difference was observed for depressed mood, anxious mood, sleep problems, daytime sleepiness, pain, and fatigue.
•Motor experiences of daily living (M-EDL): Likewise, um-PEA adjuvant therapy gave a significant reduction of UPDRS-Part II total score related to M-EDL, the scores going from 12.7 ± 1.37 at the start to 7.6 ± 1.06 at year one using um-PEA adjuvant therapy.
•Treatment with um-PEA alongside levodopa significantly reduced these symptoms: speech, dressing, turning in bed, tremors, getting out of bed, walking and balance, and freezing. Patients taking um-PEA showed a significant and progressive reduction of UPDRS-Part III total score (p<0.0001). um-PEA treatment helped most motor issues except for posture, postural tremor of the hands, kinetic tremor, and constancy of rest.
•um-PEA therapy reduced UPDRS-Part IV total score (p<0.0001) related to motor complications. The total motor complication score went from 8.8 ± 0.8 at the beginning to 4.2 ± 0.48 at 1 year using um-PEA. The authors did note that UPDRS-Part IV total score was influenced by disease duration (p= 0.0435) and dyskinesias (p=0.0001). Except for painful OFF-state dystonia, the improvement in motor complications score was statistically significant when taking um-PEA.
•The reduction of the mean score in the MDS-UPDRS sections I-IV is time-dependent and progressive for both motor and motor symptoms, supporting the notion of PEA as a disease-modifying agent.
•Using the Hoehn and Yahr (HY) Scale to determine the severity of Parkinson’s showed these changes, going from 1.8 ± 0.23 three months before the study to 2.5 ± 0.14 at the start of the clinical study and after a year of um-PEA therapy, the collective HY score was 1.9 ± 0.15.
•None of the participants reported side effects attributable to using um-PEA.
“There are no shortcuts to any place worth going.” Beverly Sills
Strengths and Weaknesses of this Study: The primary power of a prospective observational study is the accuracy of data collection concerning exposures, confounders, and endpoints. As cohort studies are observational, one of their most significant weaknesses is that people are not randomly assigned to the treatment and non-treatment groups. Furthermore, matching both groups by particular variables such as sex, age, or other confounders is not always possible. The current study was also a relatively small study limited to a specific region of Italy.
“If you have your mind in the right direction, and your heart is full of the right kind of stuff, you’ll succeed and you’ll triumph over adversity, over really anything.” Milo Ventimiglia
My Personal Experience with Palmitoylethanolamide (or PEA)*: Based on the impressive results in this clinical trial, I thought, why not try it? I am taking an ultra-micronized PEA product from Elevations Terpenes (obtained from Amazon.com). The white powder (1/2 teaspoon equal to ~800 mg um-PEA) is added to apple sauce or peanut butter, mixed up as well as possible and ingested. I have been doing this twice a day for the past six weeks. The picture below represents the need to be flexible in getting the powder suspended in the food you enjoy eating. There is not a lot of taste in the PEA powder, but you do need to ingest most of the powder somehow [If my math is correct, 100 grams of um-PEA cost ~$50 USD, and taking ~1.6 gram/day suggests this should last ~2 months and cost ~80 cents/day.].
*Medical Disclaimer: As with anything you read here, please consult your Neurologist before taking any form of palmitoylethanolamide (PEA). While it appears safe, that does not mean it is right for you to take it for your Parkinson’s.
Initial results are promising because I feel it impacts several things, including reducing generalized body pain, less daytime sleepiness, less fatigue, and better control of motor symptoms. In addition, it seems to have a calming influence on me. My golf game is still terrible, but at least I feel more stable (i.e., less shaky) hitting the ball. I realize that self-reporting is not a clinical study, and this is just the start of my evaluation of um-PEA. Likely, I will reduce the amount of um-PEA taken by half after about three months as done in the Clinical trial.
You might ask yourself, what else am I taking alongside the um-PEA? I have not changed my use of carbidopa/levodopa and the Neu-Pro patch, although some days recently, I have felt that I might need less carbidopa/levodopa with time using um-PEA. In addition, I am still taking some magnesium threonate, small amounts of vitamin B1, vitamin D3, taurine, and Ashwagandha. However, for the time being, I have stopped taking N-acetyl cysteine, glycine, niacin, alpha-lipoic acid, and acetyl-L-carnitine.
Whether these positive results are purely a placebo effect because I genuinely want this treatment strategy to work and the Italian clinical trial results were so promising, I do not know. However, I am encouraged and will continue this approach in the coming months. Long-term use of PEA in humans has shown no negative impact.
“Always seek out the seed of triumph in every adversity.” Og Mandino
Adding Luteolin to um-PEA May Further Enhance its Therapeutic Index: Several studies have added a flavonoid compound with PEA to further reduce neuroinflammation. The substance most tested was luteolin. This blog post has gone on long enough, but I wanted to at least suggest there may be some improvement in the ability of PEA to function in reducing the symptoms of Parkinson’s by adding luteolin. Stay tuned for further updates and details. When I exhaust my supply of the um-PEA powder and switch to the lower dose of PEA, I may start using a capsule form of micronized PEA with luteolin. This product would require two capsules per day to give 630 mg micronized-PEA and 30 mg of luteolin; (obtained from Amazon.com, a product of X Gold Health; ~ $37, the bottle has 120 capsules, take two capsules/day for 60 days, this would be about ~60 cents/day).
“One who gains strength by overcoming obstacles possesses the only strength which can overcome adversity.” Albert Schweitzer
Summary: In this post, I have presented an overview of inflammation/neuroinflammation, knowing that it is a big culprit in promoting the progression of Parkinson’s in many of us. Next, I reviewed the use of palmitoylethanolamide (or PEA) in reducing pain and its broad spectrum of biological actions. The use of PEA has become prominent in numerous neurodegenerative disorders.
I spent time reviewing and highlighting the remarkable clinical trial using uPEA for treating Parkinson’s, as overseen by a group of Movement Disorder physicians in Florence, Italy. While small and only a prospective observational study, this study showed significant positive changes over the year these patients took um-PEA alongside levodopa. They concluded that um-PEA has multi-functional abilities to reduce neuroinflammation. These functions make PEA a good choice for treating neurodegenerative disorders like Parkinson’s. Finally, I am jumping on this bandwagon and have started taking um-PEA with promising initial results. Stay tuned.
“I am thankful for the adversities which have crossed my pathway, for they taught me tolerance, sympathy, self-control, perseverance, and some other virtues I might never have known.” Napoleon Hill
References
Brotini, Stefania, Carlo Schievano, and Leonello Guidi. “Ultra-micronized palmitoylethanolamide: an efficacious adjuvant therapy for Parkinson’s disease.” CNS & Neurological Disorders-Drug Targets (Formerly Current Drug Targets-CNS & Neurological Disorders) 16, no. 6 (2017): 705-713.
Cordaro, Marika, Salvatore Cuzzocrea, and Rosalia Crupi. “An update of palmitoylethanolamide and luteolin effects in preclinical and clinical studies of neuroinflammatory events.” Antioxidants 9, no. 3 (2020): 216.
Landolfo, Eugenia, Debora Cutuli, Laura Petrosini, and Carlo Caltagirone. “Effects of Palmitoylethanolamide on Neurodegenerative Diseases: A Review from Rodents to Humans.” Biomolecules 12, no. 5 (2022): 667.
Beggiato, Sarah, Maria Cristina Tomasini, and Luca Ferraro. “Palmitoylethanolamide (PEA) as a potential therapeutic agent in Alzheimer’s disease.” Frontiers in pharmacology 10 (2019): 821.
Raso, Giuseppina Mattace, Roberto Russo, Antonio Calignano, and Rosaria Meli. “Palmitoylethanolamide in CNS health and disease.” Pharmacological Research 86 (2014): 32-41.
Alhouayek, Mireille, and Giulio G. Muccioli. “Harnessing the anti-inflammatory potential of palmitoylethanolamide.” Drug discovery today 19, no. 10 (2014): 1632-1639.
Petrosino, Stefania, and Aniello Schiano Moriello. “Palmitoylethanolamide: A Nutritional Approach to Keep Neuroinflammation within Physiological Boundaries—A Systematic Review.” International Journal of Molecular Sciences 21, no. 24 (2020): 9526.
Palazzo, Enza, Livio Luongo, Francesca Guida, Vito de Novellis, Serena Boccella, Claudia Cristiano, Ida Marabese, and Sabatino Maione. “Role of N-acylethanolamines in the neuroinflammation: ultramicronized palmitoylethanolamide in the relief of chronic pain and neurodegenerative diseases.” (2019): 2035-2046.
Clayton, Paul, Mariko Hill, Nathasha Bogoda, Silma Subah, and Ruchitha Venkatesh. “Palmitoylethanolamide: a natural compound for health management.” International Journal of Molecular Sciences 22, no. 10 (2021): 5305.
Rankin, Linda, and Christopher J. Fowler. “The basal pharmacology of palmitoylethanolamide.” International Journal of Molecular Sciences 21, no. 21 (2020): 7942.
Cordaro, Marika, Salvatore Cuzzocrea, and Rosalia Crupi. “An update of palmitoylethanolamide and luteolin effects in preclinical and clinical studies of neuroinflammatory events.” Antioxidants 9, no. 3 (2020): 216.
Brotini, Stefania. “Palmitoylethanolamide/Luteolin as Adjuvant Therapy to Improve an Unusual Case of Camptocormia in a Patient with Parkinson’s Disease: A Case Report.” Innovations in Clinical Neuroscience 18, no. 10-12 (2021): 12.
Siddique, Yasir Hasan. “Role of luteolin in overcoming Parkinson’s disease.” Biofactors 47, no. 2 (2021): 198-206.
Siracusa, Rosalba, Irene Paterniti, Daniela Impellizzeri, Marika Cordaro, Rosalia Crupi, Michele Navarra, Salvatore Cuzzocrea, and Emanuela Esposito. “The association of palmitoylethanolamide with luteolin decreases neuroinflammation and stimulates autophagy in Parkinson’s disease model.” CNS & Neurological Disorders-Drug Targets (Formerly Current Drug Targets-CNS & Neurological Disorders) 14, no. 10 (2015): 1350-1366.
Cover photo image by Kevin Phillips from Pixabay




Thanks again Frank for a very helpful post. The time and effort that went into this post is very much appreciated. I just read in this article https://parkinsonsnewstoday.com/news/mice-with-parkinsons-benefit-from-exercise-cholesterol-medication/?utm_source=PAR&utm_campaign=56c7fb6198-PAR_ENL_3.0_NON-US&utm_medium=email&utm_term=0_62dd4fb5e3-56c7fb6198-74271385 that the benefits of exercise in slowing the progression of PD is associated with activation of PPAR-alpha. You mentioned in your post that PEA also activates PPAR-alpha. So perhaps PEA together with exercise will have an additive effect. Also I think for activation of PPAR-alpha, PEA probably has a better safety profile than cholesterol medications. Of course whether PEA has the same efficacy and efficacy in activating PPAR-alpha as the cholesterol drugs is not known.
LikeLike
Philip, thanks for your note and comments. It would be great if exercise, together with PEA, would result in either an additive or synergistic effect to modify the progression of Parkinson’s. The problematic ‘issue’ with PEA is due to its broad range of targets, which one(s) are most relevant. And indeed, drugs have side effects, including the usually very safe anti-cholesterol compounds. I will go read this paper, sounds interesting, take care, Frank
LikeLike